- Volume 61 , Number 3
- Page: 394–7
Armadillos (Dasypus novemcinctus) as a model to test antileprosy vaccines; a preliminary report
ABSTRACT
The efficacy oftwo candidate leprosy vaccines, BCG and a mixture of BCG and killed Mycobacterium leprae, was tested in 62 armadillos caught in the wild. The abilities of the vaccines to convert lepromin-negative armadillos to a positive reaction were compared with a group of control animals. Both vaccines upgraded subsequent lepromin skin-test histopathology. The conversion results parallel the protection values obtained in some BCG vaccine trials against leprosy in humans. Before conducting expensive human trials with new antileprosy vaccines, it would be worthwhile first to evaluate them in the armadillo model.RÉSUMÉ
L'efficacité de deux candidats-vaccins anti-lèpre, le BCG et une combinaison de BCG et de Mycobacterium leprae tué, a été testée chez 62 tatous capturés dans la nature. La capacité des vaccins de convertir des tatous ne réagissant pas à la lepromine en animaux montrant une réaction positive a été comparée avec un groupe d'animaux témoins. Les deux vaccins ont modifié les caractéristiques histopathologiques d'un test cutané ultérieur à la lepromine. Les résultats de conversion sont semblables aux valeurs de protection obtenues dans certains essais de vaccination contre la lèpre chez l'homme. Il serait utile, avant de mettre en route d'onéreux essais cliniques chez l'homme avec de nouveaux vaccins anti-lèpre, de les évaluer avec le modèle du tatou.RESUMEN
Se probó la eficiencia de 2 candidatos a vacunas antileprosas, el BCG y una mezela de BCG y Mycobacterium leprae muerto, en 62 armadillos silvestres. La habilidad de las vacunas para convertir a los armadillos lepromino-negativos en lepromino-positivos se estableció por comparación con un grupo de animales control. Ambas vacunas permitieron la conversión positiva a la lepromina. En algunos estudios en humanos con la vacuna BCG se ha visto que la conversion está relacionada con protección. Se sugiere que antes de realizar costosos estudios en humanos con nuevas vacunas antileprosas, valdria la pena evaluarlas primero en el modelo del armadillo.There are now a number of candidate vaccines for leprosy, for example, a) BCG, b) BCG plus heat-killed Mycobacterium leprae, c) the ICRC bacillus, d) Mycobacterium w., and 3) M. habana. Evaluation of antileprosy vaccines is difficult. Human trials of a vaccine are expensive and require at least 10 years to obtain even preliminary results. Determining estimates of a vaccine's efficacy in an experimental animal model would be highly desirable before launching a human trial.
The growth of M. leprae in the foot pads of mice is a well established model (16). On the other hand, the lesi ons in the mouse foot pad model do not truly represent human disease, and the efficacy of a vaccine in this model is judged by the inhibition of an already limited growth of the organism (15). Mangabey monkeys are excellent models of human leprosy (l2) but are expensive and, in general, are not available in sufficient numbers for a vaccine study.
The nine-banded armadillo (Dasypus novemcinctus) develops generalized, lepromatous disease in a majority of infected animals (10). Unlike almost all of the other models for disseminated leprosy, such as the nude mouse (1,11), the T900R mouse (14), neonatally thymectomized rats (3), etc., the armadillo is immunologically intact and therefore might be expected to have the capacity to mount an effective immune response to a vaccine.
We have shown that there are armadillos which are resistant to leprosy and that the large majority of these resistant animals are lepromin skin-test positive (7). Lepromin positivity is clearly associated with resistance to leprosy in humans (2). We have evaluated two candidate leprosy vaccines in armadillos by determining their ability to convert lepromin-negative animals to lepromin skin-test positivity.
MATERIALS AND METHODS
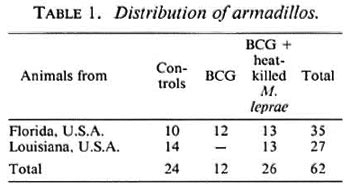
Armadillos. Sixty-two armadillos caught in the wild, weighing between 3 kg and 5 kg, and of either sex were used in this study. Thirty-five animals were obtained from the state of Florida, U.S.A., where leprosy is not found among wild armadillos. Twentyseven animals were from the state of Louisiana, U.S.A., where leprosy is endemic among wild armadillos. The Louisiana animals were not taken from areas known to be highly endemic for leprosy.
Lepromin testing and vaccinations. All of the animals were first lepromin skin-tested with heat-killed, armadillo-derived M. leprae, 1.6 × 107 in 0.1 ml of normal saline (13) injected intradermally in the abdominal skin. The injected site was identified by tattooing with India ink. At 21 days after injection the skin-test sites were biopsied using a 4-mm diameter punch.
The BCG (M. bovis, strain BCG, ATCC # 19015) was cultivated in Middlebrook 7H9 medium supplemented with albumin dextrose catalase (ADC), containing 0.05% Tween 80, in 5% C02 in humidified air at 37ºC. Subcultures were made with a 0.2-ml inoculum into 5 ml of broth, and harvested at approximately 2 weeks when the organisms had reached a concentration of approximately 1 × 108 bacilli per ml.
The animals were divided into groups of controls and those vaccinated with a) viable BCG, 1.0 x 106, b) heat-killed M. leprae, 1 x 107, or c) a mixture of viable BCG, 1.0 x 106 and heat-killed M. leprae, 1 x 107 (Table 1). All vaccinations were given intradermally in 0.1 ml of normal saline.
Three to 6 weeks after vaccination, the armadillos were again lepromin skin-tested and the skin-test sites biopsied as before.
The biopsied skin-test sites were fixed in 10% buffered formalin and processed for paraffin sections; 4-5 µm-thick sections were prepared, and for each biopsy one section was stained with hematoxylin and eosin (H&E) and one for acid-fast bacilli (AFB) (5). The lepromin skin-test biopsies were examined and classified histologically into lepromatous (LL), borderline lepromatous (BL), borderline tuberculoid (BT), and tuberculoid (TT) according to criteria which have been described previously (9).
RESULTS
The results of the lepromin skin tests are given in Table 2. For determining if an animal had acquired the ability to produce an immune granuloma in response to M. leprae, initially only BT and TT lepromin responses were considered positive, and BL and LL reactions were considered negative. By these strict criteria, when they were first tested 6 of 62 armadillos (10%) were positive and the rest were negative. The animals that were positive initially were not used in the calculation of the lepromin conversions.
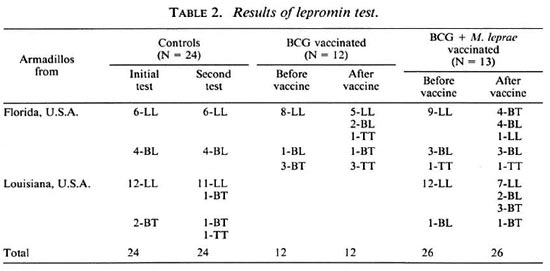
Among the 24 unvaccinated control an imals, 2 were positive initially, and of the 22 initially negative, 1 (5%) converted to positivity at the second lepromin skin test. This conversion was in a Louisiana armadillo.
In the 12 Florida armadillos vaccinated with BCG alone, 3 were initially positive; among the 9 initially negative, 2 (22%) converted to positivity. This conversion rate is not significantly different from the controls (Fisher's exact test, two-tailed, p = 0.20).
Twenty-six armadillos were vaccinated with BCG plus M. leprae. One was positive initially; in the 25 remaining, initially negative animals, 8 (32%) converted to positivity in their lepromin skin tests. This conversion rate is significantly different from that of the controls (p = 0.03) but not from that of the BCG group (p = 0.69).
Using less-stringent criteria for conversion, it may be seen in Table 2 that there was some degree of upgrading in the lepromin classification in 2 of 24 control animals, 7 of 12 receiving BCG (p = 0.003 compared to controls), and 14 of 26 armadillos receiving BCG plus M. leprae (p = 0.001 compared to controls). There is no significant difference between the animals vaccinated with BCG alone and those receiving BCG plus M. leprae even with these less-stringent criteria for conversion.
Using any degree of upgrading as a criterion, there were no statistically significant differences between the Florida and the Louisiana armadillos in the conversion of lepromin skin tests in the controls or in the animals vaccinated with BCG plus M. leprae. There was a significant difference with regard to baseline lepromin skin tests; 3 of 27 animals from Louisiana showed non-LL histology and 12 of 35 Florida armadillos did so (p = 0.041).
DISCUSSION
Tuberculoid leprosy is a self-limiting and self-healing disease. Therefore, the efficacy of a vaccine for leprosy should be assessed by its ability to protect an animal or an individual from developing lepromatous disease. Nine-banded armadillos develop generalized lepromatous leprosy in a majority of the infected animals and about 9% to 19% of them are resistant (8). Most of the resistant animals are lepromin positive (7). Even among the susceptible animals there is considerable variation in tissue response during the progress of the disease. Some animals go through a borderline phase before developing generalized lepromatous disease(6).
A positive Mitsuda reaction characterized by an epithelioid cell granuloma is a good index to assess the cell-mediated immunity response of an animal to M. leprae. Lepromin positivity, although it does not ensure complete protection from infection, indicates good resistance. Any candidate vaccine which would convert a significant majority of lepromin-negative armadillos should, in all probability, be effective in protecting the animals from lepromatous disease.
In this study using strict criteria, BCG was able to convert the lepromin skin test in 2 of 9 (22.2%) Florida armadillos. A mixture of BCG with M. leprae was able to convert 4 of 12 (33.3%) of the Florida animals and 4 of 13 (31%) of the vaccinated Louisiana animals. Although the mixture of BCG and M. leprae appears more effective than BCG alone, the difference is not statistically significant using either strict or less-stringent criteria for lepromin conversion. Further, the expected protection offered by both vaccines is only up to a maximum of 33%.
There have been several trials using BCG vaccines in humans and their results are well documented. BCG varies from 80% protection in Uganda to 20% in Burma (4). It has not been successful in preventing smear-positive leprosy in South India, where the overall efficacy was 17% to 24% (4). The results reported here on BCG-vaccinated animals more or less parallel the results reported from some human trials. This similarity suggests conducting vaccine trials using the armadillo model before embarking on time-consuming human trials.
All the 62 animals, both control and vaccinated, have now been challenged with viable M. leprae, and the results will be presented at a later date. The second generation vaccines are awaited with great interest. It may be advisable for trials of newer vaccines to be done in the armadillo model before undertaking human trials.
REFERENCES
1. COLSTON, J. J. and H ILSON, G. R. F. Growth of Mycobacterium leprae and M. marittum in congenitally athymic (nude) mice. Nature (London) 262(1976) 399-401.
2. D HARMENDRA and CHATTERJEE, B. R. Prognostic value of the lepromin test in contacts of leprosy cases. Lepr. India 27(1955)149-152.
3. FIELDSTEEL, A. H. and MCINTOSH, A. M. Effect of neonatal thymectomy and antithymocyte serum on susceptibility of rats to Mycobacterium leprae. Proc. Soc. Exp. Biol. Med. 138(1971)408-413.
4. GUPTE, M. D. Vaccines against leprosy. Indian J. Lepr. 63(1991)342-349.
5. JOB, C. K. and CHACKO, C. J. G. A modification of Fite's stain for demonstration of M. leprae in tissue sections. Indian J. Lepr. 58(1986)17-18.
6. JOB, C. K., KIRCHHEIMER, W. F. and SANCHEZ, R. M. Borderline leprosy in an experimentally infected armadillo. Int. J. Lepr. 50(1982)177-182.
7. JOB, C. K., KIRCHHEIMER, W. F. and SANCHEZ, R. M. Variable lepromin response to M. leprae in resistant armadillos. Int. J. Lepr. 51(1983)347-353.
8. JOB, C. K., SANCHEZ, R. M. and HASTINGS, R. C. Manifestations of experimental leprosy in the armadillo. Am. J. Trop. Med. Hyg. 34(1985)151-161.
9. JOB, C. K., SANCHEZ, R. M., HUNT, R. and HASTINGS, R. C. Prevalence and significance of positive Mitsuda reaction in the nine-banded armadillo. Int. J. Lepr. 55(1987)685-688.
10. KIRCHHEIMER, W. F. and STORRS, E. E. Attempts to establish the armadillo as a model for study of leprosy. 1. Report of lepromatoid leprosy in an experimentally infected armadillo. Int. J. Lepr. 39(1971)692-702.
11. KOHSAKA, K., M ORI, T. and ITO, T. Lepromatoid lesion developed in nude mice inoculated with Mycobacterium leprae. (Abstract) Int. J. Lepr. 44(1976)540.
12. MEYERS, W. M., WALSH, G. P., BROWN, H. L., BINFORD, C. H., GERONE, P. J., WOLF, R. H., GORMUS, B. J. and M ARTIN, L. Leprosy in a mangabcy monkey. (Abstract) Int. J. Lepr. 49(1981)500-502.
13. REES, R. J. W. Enhanced susceptibility of thymectomized and irradiated mice to infection with Mycobacterium leprae. Nature (London) 211(1966)657-658.
14. SENGUPTA, U. The utility of animal models for selection of a candidate vaccine for leprosy. Indian J. Lepr. 63(1991)153-158.
15. SHEPARD, C. C. The experimental disease that follows the injection of human leprosy bacilli into foot-pads of mice. J. Exp. Med. 112(1960)445-454.
16. WORLD HEALTH ORGANIZATION. Recommended safety requirements for the preparation of lepromin: a WHO memorandum. Bull. WHO 57(1979)921-923.
1. M.D., F.R.C.Path.; Laboratory Research Branch, GWL Hansen's Disease Center at Louisiana State University, P.O. Box 25072, Baton Rouge, Louisiana 70894, U.S.A.
2. B.S.; Laboratory Research Branch, GWL Hansen's Disease Center at Louisiana State University, P.O. Box 25072, Baton Rouge, Louisiana 70894, U.S.A.
3. B.S.; Laboratory Research Branch, GWL Hansen's Disease Center at Louisiana State University, P.O. Box 25072, Baton Rouge, Louisiana 70894, U.S.A.
4. Ph.D.; Laboratory Research Branch, GWL Hansen's Disease Center at Louisiana State University, P.O. Box 25072, Baton Rouge, Louisiana 70894, U.S.A.
5. M.D., Ph.D., Laboratory Research Branch, GWL Hansen's Disease Center at Louisiana State University, P.O. Box 25072, Baton Rouge, Louisiana 70894, U.S.A.
Reprint requests to Dr. C. K. Job, Consultant Pathologist, St. Thomas Hospital and Leprosy Center, Chettupattu 606801, Tamil Nadu, India.
Received for publication on 2 February 1993.
Accepted for publication in revised form on 30 April 1993.
1. Wayne M. Meyers, M.D., Ph.D., kindly served as Acting Editor in regard to the submission, review, revision, and acceptance of this manuscript.