- Volume 60 , Number 1
- Page: 100–2
Subgroups among lepromatous leprosy - a viewpoint
To the Editor:
In the recent past it has been observed that all lepromatous leprosy cases do not show uniformity in the generation of gamma-interferon in response to concanavalin A (conA), Mycobacterium leprae, and BCG (2). Similarly, in various vaccine trails nearly 30%-40% of the patients failed to show lepromin conversion (1). These parameters suggest that the lepromatous leprosy (LL) cases are immunologically heterogenous, and there is a possibility that there might be subgroups among lepromatous leprosy cases (4). In our leprosy clinic we have observed that some of the LL patients develop the LL form de novo; whereas some acquire the LL form by gradual downgrading from a higher position in the spectrum of the disease. In the present study an attempt has been made to characterize these two subgroups on the basis of the a) history of evolution of the disease, b) clinical presentation, c) bacteriological status, d) histology, and e) lepromin test.
Fifty-three patients were included in this study, 46 (86.8%) were males and seven (13.2%) females. Their ages ranged from 18 to 75 years. The lepromin test was negative in all. From the analysis of our data, two distinct patterns have emerged. In 29 patients (Group I) none had discrete lesions suggestive of a higher position in the leprosy spectrum in the past or at the time of examination. All patients showed generalized diffuse infiltration; 12 (41.4%) also had nodulation, 20 (69%) had symmetrical peripheral neuropathy, 19 (65.5%) had edema of the hands and feet, eyebrows were intact in 13 (44.8%), ichthyotic changes in skin of the lower extremities were seen in 14 (48.3%), evidence of erythema nodosum leprosum (ENL) was seen in 3 (10%), and only 3 (10%) patients had trophic ulcers. In the remaining 24 patients (Group II) history of onset of disease suggested that the initial lesion in 12 patients was an erythematous plaque, 6 had maculoanesthetic patch(es), 3 noticed symptoms of neuropathy, 2 had pain in nerve, and 1 observed nodules. On examination, in addition to lesions suggestive of lepromatous leprosy, 17 (70.8%) had a few skin lesions suggestive of a higher portion of the leprosy spectrum (BT, BB and BL), 14 (58.3%) had peripheral neuropathy, 6 (25%) had evidence of neuritis, deformities were seen in 7 (29.2%), generalized infiltration in 2 (8.3%), nodular lesions were seen in 6 (25%), eyebrows were intact in 10 (41.7%), and 6 (25%) had ENL.
The bacterial index (BI) in the slit-skin smears from four sites was uniformly high (5-6+) in Group I, and there was a good correlation with tissue BI. In Group II, the smear as well as the tissue BI showed significant variations from site to site.
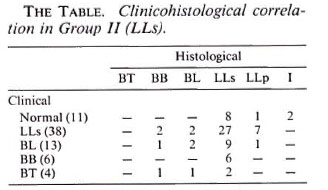
To study the nature of the cellular infiltrate, biopsies were taken from three sites. In Group I, since they had generalized diffuse infiltration, biopsies were taken from a) one of the extremities, b) the trunk, and c) apparently normal-looking skin. In Group II where there were lesions of a higher part of the leprosy spectrum, biopsies were taken from a) a lesion suggestive of lepromatous leprosy, b) a lesion suggestive of a higher part of the spectrum, and c) normal-looking skin. In Group I the tissue response in all three sites was similar and was consistent with the histology of polar lepromatous leprosy (LLp). In Group II (The Table), out of 72 biopsies, 38 were from lesions clinically suggestive of LL. Of these 38 biopsies, 27 showed histology compatible with LLs, 7 were LLp, 2 were suggestive of BL, and the remaining 2 had changes of BB. In 13 biopsies from lesions having a BL morphology, histological changes in 9 were of LLs, in 2 were of BL, and in 1 each were of LLp and BB. All six lesions with BB morphology had the histology of subpolar lepromatous leprosy (LLs). Out of 4 lesions which were clinically BT, 2 had the histology of LLs and 1 each had changes of BL and BB. Eleven biopsies were taken from apparently normal-looking skin, 8 had LLs histology, 1 had LLp histology, and 2 showed infiltrate suggestive of indeterminate leprosy (4).
From the above data it is evident that patients in Group I developed the lepromatous form de novo and should be identified as polar lepromatous (LLp). We believe that LLp cases acquire the immune defect, the activation of suppressor mechanisms or the clone deletion, from the beginning. However, with the present state of knowledge it is difficult to comment whether such an immune response is governed by some genetic factor(s) in the host variation in the strain(s) or organism, or mode of entry of the organism.
In Group II, history revealed that the initial lesions were usually of a higher part of the leprosy spectrum which gradually downgraded. These patients usually presented with lesions of lepromatous leprosy and, in addition, also showed a few lesions of a higher part of the spectrum, asymmetrical neuritis, early onset of deformities, overlapping in histology and clinical diagnosis of lesions, and variation in smear as well as in tissue BIs from site to site. These observations suggest that these cases were able to mount some resistance initially but subsequently downgraded to acquire lepromatous leprosy. We prefer to call such LL patients subpolar lepromatous (LLs).
It is well known that a percentage of lepromatous patients show upgrading with treatment and more frequently on administration of immunotherapy (1). In our earlier study, where oral zinc was tried as an immunomodulator, we failed to observe histological upgrading as well as lepromin conversion in LLp patients, while a small percentage of LLs patients did show the upgrading phenomenon (3). These observations indicate that LLs patients are absolutely unresponsive.
In conclusion, we would like to stress that LLp and LLs patients have some basic difference(s) in pathogenesis. These two subtypes can be easily differentiated on the basis of clinical presentation, the BI, and the nature of the infiltrate. The in vitro tests (LLT and LMIT) may not be reliable parameters since they, like lepromin, can be influenced by the degree of immunosuppression and bacterial load. This subgrouping of lepromatous leprosy cases can be useful in assessing the extent of benefits of various vaccines, immunomodulators, interpretations of immunological parameters, and maybe in the understanding of the pathogenesis of leprosy.
- Narendra K. Mathur, M.D.
Professor and Head
- Dinesh C. Mathur, M.D.
Rajesh D. Mehta, M.D.
Asit Mittal, M.D.
Suresh K. Jain, M.D.
Department of Dermatology
S.M.S. Medical College and Hospital
Jaipur 302004, India
- B. C. Sangal, M.D.
Department of Pathology
S.D.M. Hospital and Research Center
Jaipur 302015, India
Acknowledgment. The authors wish to acknowledge financial grant no. SP/SO/B-30/87 from the Department of Science and Technology, India, for the support of the work published here.
REFERENCES
1. ANTIA, N. H. Leprosy vaccine-a reappraisal. (Editorial) Int. J. Lepr. 56(1988)310-313.
2. KAPLAN, G., WEINSTEIN, D. E., STEINMAN, R. M., LEVIS, W. R., ELVERS, U., PATARROYO. M. E. and COHN, Z. A. An analysis of in vitro T cell unresponsiveness in lepromatous leprosy. J. Exp. Med. 162(1985)917-929.
3. MATHUR, N. K., BUMB, R. A., MANGAL, H. N. and SHARMA. M. L. Oral zinc as an adjunct to dapsone in lepromatous leprosy. Int. J. Lepr. 52(1984)331-338.
4. RIDLEY, D. S. Histological classification and immunological spectrum of leprosy. Bull. WHO 51(1974)451-465.
Reprint requests to Dr. N. K. Mathur, C-24 Peeyush Path, Bapu Nagar, Jaipur 302004, India.