- Volume 59 , Number 4
- Page: 537–47
Impairment of fingertip vasomotor reflexes in leprosy patients and apparently healthy contacts
ABSTRACT
Fingertip blood-flow velocity and its control by vasomotor reflexes were studied in leprosy patients and in healthy controls with a laser Doppler flowmeter. In newly registered patients, the flow was significantly lower than in the healthy controls, and even lower values were recorded in the longstanding patients with lower limb ulcers and/or deformity. The newly registered patients showed substantially impaired vasomotor reflex responses in the fingertips to cold challenge of the opposite hand or deep inspiratory gasp. Low blood flow and impairment of vasomotor reflexes were more prominent in those leprosy patients who showed clinical evidence of neuropathy and/or histological evidence of reaction in a punch biopsy of leprosy skin lesions. This aspect of dysautonomia to cold challenge was particularly prominent in apparently healthy, fully treated ex-patients. There was an unexpectedly high prevalence of impairment of vasomotor reflexes in newly registered and apparently healthy, adequately treated leprosy patients. The method is very sensitive, and it remains to be established whether the lesions it detects are nonprogressive residues, or previous nerve damage, or an indication of on-going nerve damage. A minority of leprosy contacts showed impairment of vasomotor reflexes. Those with two or more affected fingers were more likely to have had a higher level of exposure to Mycobacterium leprae than those with one or no affected fingers. The cause of this unexpected impairment of fingertip vasomotor reflexes in a minority of leprosy control workers has not yet been determined.RÉSUMÉ
La rapidité du flux sanguin au bout des doigts et son contrôle par des réflexes vasomotcurs ont été étudiés chez des malades de la lèpre et des témoins en bonne santé avec un appareil à la ser de mesure de l'effet Doppler. Chez les patients nouvellement enregistrés, le flux était significativement plus lent que chez les témoins en bonne santé, et des valeurs encore plus basses ont été notées chez des patients de longue date présentant de ulcères et/ou déformités des membres inférieurs. Les patients nouvellement enregistrés montraient des réponses réflexes considérablement amoindries aux bouts de doigts quand la main opposée était soumise au froid ou lors de l'inspiration profonde. La faiblesse du flux sanguin et la détérioration des réflexes vasomotcurs étaient plus importantes chez les malades de la lèpre qui montraient des signes cliniques de neuropathic et/ou des signes histologiques de réaction au niveau d'une biopsie de lésions cutanées. Cet aspect de dysautonomie vis-à-vis du froid était particulièrement important chez des anciens patients ayant terminé leur traitmeent et apparemment en bonne santé. Il y avait une prevalence plus haute qu'attendue de détérioration des réflexes vasomotcurs chez les malades de la lèpre nouvellement enregistrées, apparemment en bonne santé, et traités de manière adéquate. La méthode est très senscible, et il reste à établir su les lésions qu'elle détecte sont des séquelles qui n'évoluent plus, c'est-à-dire des lésions nerveuses antérieures, ou indiquent une détérioration nerveuse encore en progrès. Une minorité des contacts de malades de la lèpre montrait une altération des réflexes vasomotcurs. Ceux qui présentaient deux doigts affectés ou plus avaient plus vraisemblablement été exposés de manière plus importante à Mycobacterium leprae que ceux qui n'avaient pas de doigt affecté, ou n'en avaient qu'un. La cause de cette détérioration inattendue des réflexes vasomotcurs des bouts des doigts chez une minorité de travailleurs de la lèpre n'a pas encore été déterminée.RESUMEN
Utilizando un flujómetro Doppler de rayo laser, se estudió la velocidad del flujo sanguíneo en la punta de los dedos y su control por reflejos vasomotores, en pacientes con lepra y en individuos sanos. El flujo sanguíneo fue significativamente menor en los pacientes recién registrados que en los controles sanos y fue todavía menor en los pacientes con lepra de muchos años y úlceras o deformaciones en los miembros inferiores. Los pacientes recién registrados mostraron respuestas reflejas vasomotoras substancialmente disminuidas cuando se retaron con estimulos fríos en la mano opuesta o cuando se sometieron a esfuerzos inspiratorios profundos. La baja velocidad de flujo sanguíneo y la afectación de los reflejos vasomotores fueron más prominentes en aquellos pacientes con evidencias clínicas de neuropatía o con evidencias histológicas de reacción en biópsias de lesiones dérmicas. Este fenómeno de disautonomía al reto con frío fue particularmente prominente en ex-pacientes completamente tratados y aparentemente sanos. La incidencia de alteraciones en los reflejos vasomotores de los pacientes recién registrados y en los pacientes adecuadamente tratados y aparentemente sanos resultó inesperadamente elevada. El método es muy sensible pero queda por establecerse si las lesiones que detecta son residuales y no progresivas o si son indicativas de un daño nervioso en progreso. Una minoría de los contactos de los pacientes con lepra mostraron un abatimiento de sus reflejos vasomotores. Es muy probable que los pacientes con 2 o más dedos afectados hayan tenido mayor grado de exposición al Mycobacterium leprae que los pacientes con un solo dedo afectado o que aquellos sin afección digital. La causa de la inesperada alteración de los reflejos vasomotores en una minoría de los contactos clínicamente sanos no se ha determinado.Peripheral neuropathy is a common and serious complication of leprosy, and it is the basis of much of the deformity and disability associated with the disease (22). The onset may be marked by swelling and tenderness of the peripheral nerves or by signs and symptoms of peripheral neuritis, indicating the need for anti-inflammatory treatment to halt its course and prevent permanent damage to the affected nerves (12). The neuropathy is often clinically silent in its evolution, so that early diagnosis may be difficult and permanent nerve damage often cannot be prevented, even by careful clinical management (13).
The damage in leprosy neuropathy is well recognized in the somatic motor and sensory pathways in the nerves (5, 7), but autonomic function can also be disturbed(12, 16, 17). We have recently developed tests for the detection of impairment of vasomotor reflexes controlling the blood flow at the fingertips (2). The new method appeared to be sensitive for detecting minor abnormalities, and it gave reproducible results. The measuring head of the laser Doppler flowmeter is small (3 mm in diameter), allowing the responsiveness of skin vessels in individual fingertips to be assessed, and so the new method may be particularly appropriate for the investigation of leprosy patients. In preliminary studies on small groups of subjects, the detection of impaired vasomotor reflexes clearly distinguished newly registered leprosy patients from healthy people (2).
This paper reports our findings with the new technique on the prevalence of impairment of vasomotor reflexes in various groups of leprosy patients and in apparently healthy people, with differing degrees of contact with such patients, to assess the potential value of tests for the impairment of vasomotor reflexes in the diagnosis of leprosy neuropathy.
MATERIALS AND METHODS
Subjects
The study was carried out at the Richardson Leprosy Hospital (RLH), Miraj, Maharashtra, India, with approval from the local Ethics Committee. All participants had given informed consent without coercion. The following groups of leprosy patients were studied:
Newly registered leprosy patients. Thirty-eight outpatients attending RLH over a period of 12 weeks were recruited. No attempt was made to select on any basis other than willingness to participate. There were 32 men and 6 women ranging in age from 16 to 71 (mean 39.4, S.D. 15.8). Histopathological diagnosis based on a punch biopsy from the edge of a lesion showed that their classifications on the Ridley-Jopling(25) scale was 13 borderline tuberculoid (BT), 12 borderline lepromatous (BL), 7 lepromatous (LL) and 5 indeterminate. One patient's biopsy was not diagnostic of leprosy, but clinical assessment was indicative of early disease.
Careful clinical examination was made by the diagnosing doctor and the assessing physiotherapist to determine whether the patient showed evidence of impairment of the various modes of somatic sensation and/or motor impairment. Sensation in the upper limb was examined for integrity of touch (with von Frey hairs) (19) and by pin prick. The median and ulnar nerves were palpated for enlargement. The physiotherapist assessed the strength of the various muscle groups in the upper limb. From this examination, the subjects were classified as having "substantial" peripheral neuropathy if there were widespread abnormalities in the various groups of tests, and as "partial" if there was either a) focal partial loss of sensation, or b) a palpably swollen nerve, or c) convincing evidence of muscle weakness in some muscle groups.
Histological evidence of reversal reaction was sought in the diagnostic punch biopsies of the leprosy skin lesion.
Longstanding leprosy patients with orthopedic problems. Eleven male patients (mean age 33.4, S.D. 12.1; range 20-55 years) with longstanding disease receiving inpatient treatment in the Medical Ward at RLH for foot/leg ulcers or serious deformity were tested.
Known treated ex-patients. Twelve men (mean age 33.3, S.D. 9.8; range 22-51 years) currently employed in RLH and free from obvious clinical stigmata were recruited. Each person had indicated in a confidential interview that, in the past, he had completed the full course of treatment.
Controls
Two groups of healthy subjects were studied:
Healthy controls (Miraj). Thirty-five employees and visiting workers at RLH (mean age 35.7, S.D. 10.0; range 20-61 years). There were 29 men and 6 women (28 Indians, 7 Europeans). All appeared healthy and denied having had leprosy in the past.
Unexposed healthy controls (Dundee). Fourteen healthy European volunteers were studied in Dundee (mean age 33.7, S.D. 12.0; range 23-60 years). There were seven men and seven women in this group. They were studied in an environmentally controlled room in Dundee under ambient temperature conditions similar to those in Miraj.
Experimental protocol
Each subject was seated comfortably with the forearm and hand resting on a table at heart level at an ambient temperature of 26-29ºC. These conditions would be expected to induce near maximal peripheral vasodilatation and a stable blood flow through the fingertips in healthy subjects. Each subject was allowed to equilibrate under these conditions for at least 15 min before measurements were started. All four fingers were studied on one hand before the other hand was tested; no measurements were made on the thumbs.
Measurement of blood flow
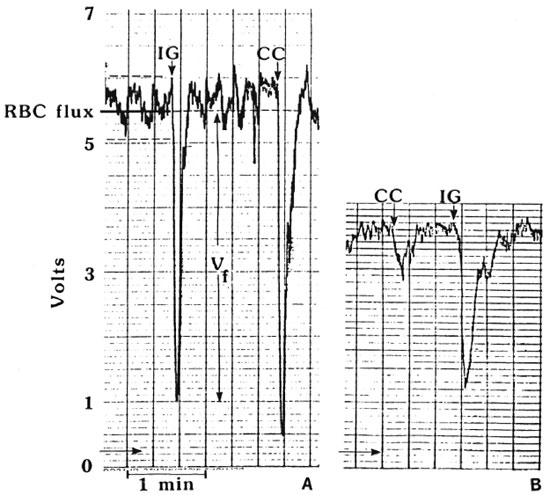
A laser Doppler flowmeter (model PF2; Perimed, Stockholm, Sweden) was used to measure the blood flow through the skin over the pulp of the distal phalanx with machine settings "gain 3; band width 12 kHz; time constant 0.2 s; artifact filter off." This machine measures the movement of erythrocytes in the most superficial 1 mm of skin from changes in the wavelength of light reflected in the tissue from the beam of coherent light emitted by the laser. The size of the signal recorded from the flowmeter (red blood cell or RBC flux, expressed in volts) is directly related to the product of the number of moving cells and their mean velocity (11, 20, 21). The fiber-optic sensor was attached to the fingertip by a probe holder fixed with double-sided adhesive tape ensuring optimal alignment between sensor head and skin surface. The output signal was recorded continuously during each experiment on a chart recorder (SE 120; BBC Goetz Mctrawatt, Austria) calibrated to a full-scale deflection of 10 V. The pulp of the fingertip was selected for investigation because its abundant arterio-venous anastomoses are under strict autonomic control(9,10). The "resting" fingertip blood flow was taken as the mean RBC flux value during a 3-min observation period on an equilibrated subject. Figure 1 compares a tracing from a normal subject with that from a newly registered leprosy patient who had a reduced steady-state fingertip blood flow.
Fig. 1. Tracings of RBC flux from (A) healthy Dundee adult and (B) leprosy patient. Under steady-state conditions, the RBC flux is pulsatile (in time with the arterial pulse): the mean value is recorded for subsequent analysis. RBC flux is lower in the leprosy patient (3.8 V) than in the healthy subject (5.8 V). Responses to cold challenge (CC) and deep inspiratory gasp (IG) are recorded as amplitude (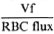 ). Leprosy subject illustrated here showed marked impairment of CC.
). Leprosy subject illustrated here showed marked impairment of CC.
Elicitation of vasomotor reflexes
The autonomic control of fingertip blood flow was assessed by attempting to elicit two simple vasomotor reflexes. The "inspiratory gasp" reflex was induced by the subject taking a sudden deep full inspiration and holding it for 10 sec, or until minimal blood flow was recorded. In a normal subject there was a short lag period (2 to 3 sec) before the RBC flux fell sharply, and this was followed by a quick return to the original level. The "cold challenge" was provoked by quickly cooling the contralateral hand by immersion in a bath of cold water (at or just below 15ºC). The water was stirred vigorously to ensure a rapid fall in the temperature of the hand skin. The details of the experimental methods and their physiological validation have been published previously (2). To minimize variability in the gasp reflex due to a misunderstanding from language difficulties, up to three deep gasps were elicited for tests on each finger and the largest response was used for subsequent analysis. During the cold challenge tests, the contralateral hand was dried and allowed to reheat before testing the response on another finger. From each test tracing, the "amplitude" (Vf/RBCmax) of reflex slowing of blood flow as recorded (Fig. 1), since our previous studies had shown that this feature of the response was the most reproducible parameter and gave the greatest discrimination between leprosy patients and healthy people(2).
Statistical interpretation of results
The differences in the magnitude of resting blood flow or in the extent of the response to inspiratory gasp or cold challenge in the various groups of subjects were sought by χ2 tests and one-way analysis of variance with follow-up analysis using t tests.
It has been shown previously (2) that there was no statistically significant difference between 10 healthy subjects in Dundee and 10 healthy Indian subjects in either the RBC flux or in the percentage fall induced by forced inspiration or by immersion of the opposite hand in cold water. Furthermore, these results were closely similar to those reported in a further group of healthy Dundee subjects (15). Accordingly, these results were combined to calculate the reference range for each parameter (3). The lower limits were 3.04 V for RBC flux, 59.9% fall for inspiratory gasp, and 48.3% fall for cold challenge. With this background, the results on each finger can be classified as "impaired" if they are less than the lower limit of the reference range. The 95% confidence interval (CI) for each estimate of proportion of subjects with abnormality was taken from standard tables (7).
The calculations were performed on a microcomputer with the Statgraphics package (Release 3.0; STSC Inc., Rockville, Maryland, U.S.A.).
RESULTS
Leprosy patients
Table 1 summarizes the results on steady-state fingertip blood flow and the amplitude of the responses in vasomotor reflexes to cold challenge and inspiratory gasp.
Fingertip blood flow. The RBC flux measurements were highest in the group of unexposed healthy controls living in Dundee, and significantly lower (p < 0.05) in the healthy contacts in Miraj, the newly diagnosed leprosy patients, the treated ex-patients, and the longstanding patients with orthopedic problems. The mean levels for all of these groups of leprosy patients were significantly different from each other (p < 0.05).
Fingertip vasomotor reflexes. The amplitude of the reflex vasoconstriction induced by inspiratory gasp (IG) was significantly greater than that induced by cold challenge (CC) to the contralateral hand in all groups (p < 0.05). The mean IG results in the newly registered patients and the treated ex-patients were significantly lower than those of the unexposed healthy controls and the healthy Miraj contacts, but the two patient groups did not differ significantly from each other. The mean falls in amplitude of IG and CC were significantly lower in newly registered patients than in either of the two groups of controls. The mean amplitude of fall in CC was significantly lower (p < 0.05) in the ex-patients than in the newly registered patients. The resting RBC flux measurements in the longstanding leprosy patients were generally so low that it was not possible to make meaningful assessments of their responses to the vasomotor reflex tests, but most of their fingers can be regarded as nonresponsive.
Proportion of fingers within individual subjects' hands with reduction in RBC flux and impairment of CC and IG reflexes in each clinical group. This is a measure of the extent of involvement in each patient's hand which can range from no finger involvement (0%) to defects in all eight fingers (100%). Table 1 shows the mean ± S.D. for these measurements for the extent of involvement in the individual subjects in the various clinical groups. The unexposed healthy subjects in Dundee and the healthy contacts in Miraj had hands with very few fingers with reduced RBC flux, or impaired IG or CC responses. In the hands of the newly registered patients, approximately half the fingers had reduced blood flow and impaired IG or CC vasomotor reflexes. By contrast, the hands of the treated ex-patients showed relatively few fingers with reduced RBC flux but fingers with impaired IG were common, and most hands had some fingers with reduced CC.
Table 2 summarizes the inter-relationship between the abnormalities of the vasomotor reflexes. The abnormalities are presented in increasing severity from "cold-impaired, gasp-unaffected" on the left to "cold-absent, gasp-absent" on the right. The differences between groups are highly significant (χ2 = 240.9; p < 0.00001). The prevalence of impairment or absence of either reflex was seen in 6.25% of the Dundee normals [95% confidence interval (CI )2.58%-12.74%]. For the healthy Miraj contacts the corresponding figures were 19.49% (95% CI 14.88%-25.08%). The observed prevalence of abnormality was 55.93% in the newly registered patients (95% CI 49.60%-62.22%) and 79.35% in the treated ex-patients (95% CI 69.64%-87.06%). The concurrent absence of IG and CC was the most common combination in the newly registered patients; whereas the treated ex-patients most commonly showed an absence of CC with lesser abnormality in the IG response.
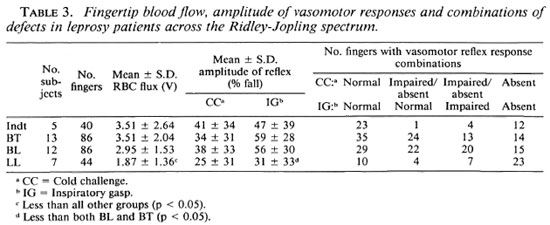
Relationship of vasomotor impairment to features of leprosy. The findings in the newly registered patients are summarized in Table 3. The abnormalities appeared to be least common in the patients with indeterminate leprosy and progressively more common and severe as the spectrum is crossed from BT through BL to LL leprosy (χ2 = 52.17; p < 0.0001). The proportion of fingers with impairment of either or both vasomotor reflexes was significantly greater than that seen in the healthy contacts in all groups of leprosy patients (p < 0.001).
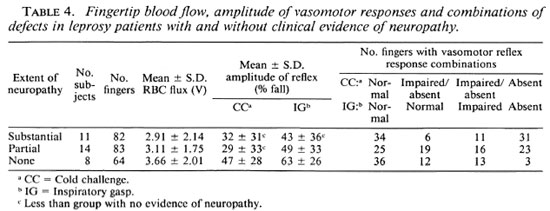
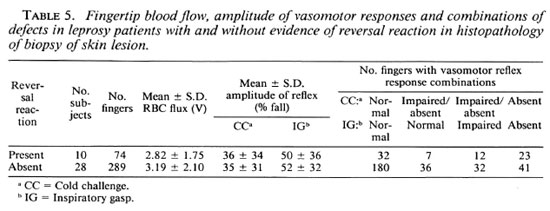
The vasomotor reflex responses were significantly reduced (p < 0.05) in those patients with either partial or substantial clinical evidence of peripheral neuropathy (Table 4). Table 5 shows that the vasomotor reflex responses were lower in the fingers of those patients whose skin biopsies showed evidence of reversal reactions (42 of 74 fingers examined, 56.8%) than in the patients with no histological evidence of reversal reaction (109 of 289, 37.7% of fingers examined; χ2 = 8.03; p < 0.005).
Healthy contacts
The distribution of amplitude of vasomotor reflexes, both CC and IG, in the healthy contacts was generally similar to that seen in the Dundee controls but was more severely skewed in the contacts, particularly in the CC reflexes (Fig. 2). In an attempt to determine the basis of this discrepancy, those contacts with more than one finger showing impairment of either the CC or IG response (the "impaired") were compared with the remainder of the healthy contacts (the "unimpaired"). The subjects with impaired responses were all ethnic Indians (mean age 37.5 years, S.D. 7.23); ten were employed in leprosy hospitals and two were family contacts of patients. In order to assess the relative intensity of contact with leprosy patients, the healthy contacts were scored on a five-point scale: 1 = short-term visitors to RLH or others with minimal contact; 2 = students in a training course for paramedical workers; 3 = ancillary workers in RLH; 4 = paramedical workers of long standing (5 years); 5 = doctors, nurses or family members of patients. Figure 3 compares the intensity of contact in the "impaired" with that recorded for the "unimpaired." Mann-Whitney testing showed that the "impaired" scored higher than the "unimpaired" (p = 0.05).
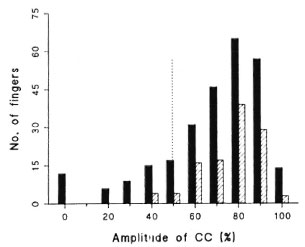
Fig. 2. Distribution of amplitude of cold challenge (CC) in fingertip skin of unexposed healthy European adults ( ) and apparently healthy contacts of leprosy patients (
) and apparently healthy contacts of leprosy patients ( ). Lower limit of reference range is indicated by vertical dotted line.
). Lower limit of reference range is indicated by vertical dotted line.
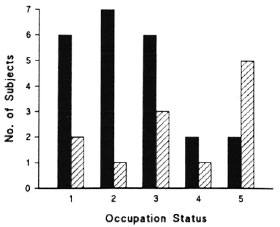
Fig. 3. Occupational status of healthy contacts of leprosy patients (ranked in anticipated increasing intensity of exposure); ( ) = those with one or no fingers showing impairment of inspiratory gasp (IG) or cold challenge (CC) reflexes; (
) = those with one or no fingers showing impairment of inspiratory gasp (IG) or cold challenge (CC) reflexes; ( ) = those with two or more fingers showing impaired IG or CC reflexes.
) = those with two or more fingers showing impaired IG or CC reflexes.
DISCUSSION
Leprosy patients
We have shown that the fingertip blood flow is reduced and that there is impaired vasomotor control in substantial numbers of patients with active or apparently successfully treated leprosy. In patients with active disease, the abnormalities are more severe at the lepromatous end of the spectrum; those showing clinical evidence of neuropathy or histological evidence of reversal reaction had greater abnormality. The ultimate cause of the reduced blood flow in the fingertip skin of leprosy patients is clearly a consequence of failure of the arteriolar system to permit a high perfusion rate in the microcirculation at high environmental temperatures-a physiological adaptation that would be expected to produce marked peripheral vasodilatation. It is not yet known whether this is a consequence of increased viscosity of the blood (31) flowing through dilated arterioles, of reduced arterial inflow (14) to the hands possibly due to tapering disease of the digital arteries (6, 33) , or of an abnormality in physiological control of the arterioles resulting in physiologically inappropriate tonic spasm of these vessels. It is nevertheless noteworthy that lepromatous leprosy patients more commonly showed abnormalities in larger arteries (4) and veins (1) than did tuberculoid patients. We have recently shown that this decrease in blood flow results in the patients showing inappropriately cold fingers (unpublished data) at high ambient temperatures.
We have shown previously (2) that both the afferent and efferent pathways of the vasomotor reflexes are involved, but that the damage to the latter is predominant. Vasomotor reflex impairment appears to be more frequent than the traditionally sought clinical evidence of somatic neuropathy, such as loss of motor power or sensation, but this may not be too surprising since many untreated patients have substantial histological damage demonstrable in nerve biopsies (24) and normal large-fiber conduction can be recorded with C and Ad fiber damage (8). The relatively early appearance of autonomic impairment may be due to the efferent pathway for these reflexes passing through the unmyelinated nerve fibers. There is now evidence that Mycobacterium leprae has a predilection for early invasion of small or unmyelinated fibers (18, 32) although all classes of fibers can be damaged in advanced disease (23). Shetty, et al. (30) have shown that the earliest changes in teased nerve preparations affect the unmyelinated fibers. Moreover early abnormalities are present in the dermal nerves distant from the skin lesions (28).
The clinical relevance of knowledge of impairment of the fingertip vasomotor reflexes has not yet been evaluated, but the presence of the abnormality is a clear indication that leprosy neuropathy is damaging the peripheral nerves. Since leprosy neuropathy is usually a progressive condition with interstitial fibrosis causing irreversible damage (13), the simple tests reported in this paper may have the potential for diagnosis of neuropathy before irreversible damage is done and while anti-inflammatory treatment can reduce edema and intraneural pressure to allow regeneration to take place.
Healthy contacts
This study has shown that the response to cold challenge is more frequently impaired in the contacts of leprosy patients than in unexposed healthy people, but that blood flow is unimpaired and the response to inspiratory gasp is normal in most leprosy contacts. It is not yet possible to determine whether the abnormalities detected by us in leprosy contacts result from damage to the cold afferent fibers or to the autonomic efferent fibers. Since the abnormality is more frequent in contacts with the greatest intensity of potential exposure, such as doctors, nurses or household contacts, it is possible that at least some of the subjects with impaired vasomotor reflexes have pre-symptomatic disease as has been reported by others (26-29).
There is, however, a problem in the interpretation of our findings in apparently healthy leprosy control workers for whom the risk of infection is small. We hope in future studies to eliminate the unlikely probability that vasomotor reflex impairment in contacts is due to early infection from frequent contact with patients with active disease, and to investigate whether dysautonomia is a) an immunopathological response to heavy exposure to M. leprae, such as an immunological hypersensitivity-induced noninfective neuropathy; or b) a residue of previous successfully treated leprosy. We favor the last explanation since leprosy regretfully still carries serious social stigma in endemic regions, and so there is a great incentive for treated patients to conceal a history of the disease. It is important to elucidate this problem since we cannot yet discount the possibility that there may be a serious long-term risk to leprosy paramedical workers.
Acknowledgment. We are grateful to LEPRA for generous financial support and to Dr. Waters of The Leprosy Mission who made possible the visit of NCA and JSB to the Richardson Leprosy Hospital, Miraj, Maharashtra, India. We wish to acknowledge the help and support we received from Dr. D. Jadhav and the many staff members at Richardson Leprosy Hospital whose help made this study possible. We have also had helpful discussion with Dr. W. C. S. Smith. The study would not have been possible but for the active cooperation of the many patients who participated in the study.
REFERENCES
1. BANSAL, R., KAUR, S., KUMAR, B., SHARMA, V. K., KATARIYA, S., CHAKRAVARTI, R. N. and BUSHARNAMATH, S. R. Venous involvement in leprosy: a venographic and histopathologic correlation. Int. J. Lepr. 55(1987)499-506.
2. BECK, J. S., ABBOT, N. C, SAMSON, P. D., BUTLIN, R., GRANGE, J. M., CREE, I. A., FORSTER, A. and KHAN, F. Impairment of vasomotor reflexes in the fingertips of leprosy patients. J. Neurol. Neurosurg. Psychiat. 1991 (in press).
3. BROWN, R. A. and BECK, J. S. Statistics on microcomputers. A non-algebraic guide to the appropriate use of statistical packages in biomedical research and pathology laboratory practice. 6. Statistical methods for diagnostic tests. J. Clin. Pathol. 42(1989)225-230.
4. CHOPRA, J. S., KAUR, S., MURTHV, J. M., KUMAR, B., RADHAKRISHNAN, K., SURI, S. and SAWHNEY, B. B. Vascular changes in leprosy and their role in the pathogenesis of leprosy neuritis. Lepr. India 53(1981)443-453.
5. DASH, M. S. A study of the conduction velocity of sensory fibers of the ulnar nerve in leprosy. Int. J. Lepr. 35(1967)460-469.
6. DEBI, B. P., MOHANTI, H. C, TRIPATHY, N., TOMPE, D. B. and SARANGI, B. K. Arteriographie patterns of plantar ulcers in lepromatous leprosy-a study of 20 cases. Lepr. India 52(1980)429-432.
7. DIEM, K. Documenta Geigy: Scientific Tables. 6th ed. Manchester: Geigy Pharmaceutical Co., Ltd., 1962, pp. 85-103.
8. DONDE, S. V., SHAH, A. and ANTIA, N. H. Nerve conduction in leprosy: in vivo and in vitro study. Lepr. India 55(1983)12-21.
9. GRANT, R. T. and BLAND, E. F. Observation on arteriovenous anastomoses in human skin and in the bird foot with special reference to cold. Heart 15(1931)385-406.
10. HALES, J. R., IRIKI, M., TSUCHIYA, K. and KOZAWA, E. Thermally induced cutaneous sympathetic activity related to blood flow through capillaries and arteriovenous anastomoses. Plfuegers Arch. 375(1978)17-24.
11. HOLLOWAY, G. A. Laser Doppler measurement of cutaneous blood flow. In: Non-Invasive Physiological Measurement, Vol. 2. Rolfe, P., ed. London: Academic Press, 1983, pp. 219-249.
12. JACOBSON, R. R. Treatment. In: Leprosy. Hastings, R. C, ed. Edinburgh: Churchill Livingstone, 1985, pp. 53-87.
13. JOB, C. K. Nerve damage in leprosy. Int. J. Lepr. 57 (1989) 532-539.
14. JOHNSON, A. C, REDDY, R., JOHNSON, S. and JAMES, A. E. Lower limb angiography in leprosy. Radiology 126(1978)327-332.
15. KHAN, R., SPENCE, V. A., WILSON, S. B. and ABBOT, N. C. Quantification of sympathetic vascular responses in skin by laser Doppler flowmetry. Int. J. Microcirc. Clin. Exp. 10(1991)145-153.
16. KYRIAKIDIS, M. K., NOUTSIS, C. G., ROBINSON-KYRIAKIDIS, C. A., VENETSIANOS, P. J., VYSSOULIS, G. P., TOUTOUZAS, P. C, PARISSIS, N. G. and AVGOUSTAKIS, D. G. Autonomic neuropathy in leprosy. Int. J. Lepr. 51(1983)331-335.
17. MCLEOD, J. G., HARGRAVE, J. C, WALSH, J. C, BOOTH, G. C, GYE, R. S. and BARON, S. A. Nerve conduction studies in leprosy. Int. J. Lepr. 43(1975)21-31.
18. MEHTA, L., SHETTY, V. and ANTIA, N. H. Study of early nerve lesions in mice infected with M. leprae. Lepr. India 48(1976)31-35.
19. NAAFS, B. and DAGNE, T. Sensory testing; a sensitive method in the follow-up of nerve involvement. Int. J. Lepr. 45(1977)364-368.
20. NILSSON, G. E., TENLAND, T. and OBERG, P. A. A new instrument for continuous measurement of tissue blood flow by light beating spectroscopy. IEEE Trans. Biomed. Eng. 27(1980)12-19.
21. NILSSON, G. E., TENLAND, T. and OBERG, P. A. Evaluation of a laser Doppler flowmeter for measurement of tissue blood flow. IEEE Trans. Biomed. Eng. 27(1980)597-604.
22. PFLATZGRAFF, R. E. and BRYCESON, A. Clinical leprosy. In: Leprosy. Hastings, R. C , ed. Edinburgh: Churchill Livingstone, 1985, pp. 134-176.
23. PANDYA, S. S. and CHULAWALA, R. G. Electrophysiologic and histopathologic studies in leprosy and some acrodystrophic neuropathies. Int. J. Lepr. 49(1981)398-405.
24 PEDLEY, J. C, HARMAN, D. J., WAUDBY, N. and MCDOUGALL, A. C. Leprosy in peripheral nerves: histopathological findings in 119 untreated patients in Nepal. J. Neurol. Neurosurg. Psychiat. 43(1980)198-204.
25 RIDLEY, D. S. and JOPLING, W . H. Classification of leprosy according to immunity; a five-group system. Int. J. Lepr. 34(1966)255-273.
26. SAMPATAVANICH, S., SAMPOONACHOT, P., KONGSUEBCHART, K., RAMASOOTA, T., PlNRAT, U., MONGKOLWONGROI, P., OZAQA, T., SASAKI, N. and ABE, M. Immunoepidemiological studies on subclinical infection among leprosy household contacts in Thailand. Int. J. Lepr. 57( 1989)752-765.
27. SEHGAL, V. N. Significance of the local sweat response in the diagnosis of leprosy. Dermatológica 148(1974)217-223.
28. SHETTY, V. P., ANTIA, N. H. and JACOBS, J. M. The pathology of early leprous neuropathy. J. Neurol. Sci. 88(1988)115-131.
29. SHETTY, V. P., MEHTA, L. N., ANTIA, N. H. and IRANI, P. F. Teased fibre study of early nerve lesions in leprosy and in contacts, with electrophysiological correlates. J. Neurol. Ncurosurg. Psychiat. 40(1977)708-711.
30. SHETTY, V. P., MEHTA, L. N., IRANI, P. F. and ANTIA, N. H. Study of the evolution of nerve damage in leprosy. Part I. -Lesions of the index branch of the radial cutaneous nerve in early leprosy. Lepr. India 52(1980)5-18.
30. SIMPSON, L. D. Altered blood rheology in the pathogenesis of diabetic and other neuropathies. Muscle Nerve 11(1988)725-744.
31. WEDDELL, G. Disorders of peripheral cutaneous nerves. J. Invest. Dermatol. 69(1977)130-135.
32. YADAV, S. S. Artériographie evaluation of vascular changes in leprosy. Angiology 29( 1978)17-21.
1. M.Sc, Research Assistant, University of Dundee, Dundee, U.K.
2. M.D., F.R.C.Path., F.R.S.E., Professor, University of Dundee, Dundee, U.K.
3. M.B., Ph.D., M.R.C.Path., Senior Lecturer, Department of Pathology, University of Dundee, Dundee, U.K.
4. B.Sc, Ph.D., Senior Lecturer, Department of Mathematical Science, University of Dundee, Dundee, U.K.
5. M.B., M.R.C.P., Consultant Neurophysiologist, University of Dundee, Dundee, U.K.
6. M.B., M.P.H., Superintendent, Richardson Leprosy Hospital, Miraj, Dist. Sangli, Maharashtra, India.
7. M.B.B.S., Medical Officer, Richardson Leprosy Hospital, Miraj, Dist. Sangli, Maharashtra, India.
8. M.D., M.Sc, Reader, Department of Microbiology, National Heart and Lung Institute, London, U.K.
Reprint requests to Prof. J. Swanson Beck, Pathology Department, Ninewells Hospital and Medical School, Dundee DDI 9SY, U.K.
Received for publication on 28 May 1991.
Accepted for publication on 24 June 1991.