- Volume 58 , Number 2
- Page: 328–33
Rapid serodiagnosis for leprosy - a preliminary study on latex agglutination test
ABSTRACT
In this study, we have developed two latex agglutination tests (LATs) with phenolic glycolipid-I (PGL-I) and natural disaccha-ride-octyl-bovine serum albumin (ND-O-BSA) as antigens in 110 leprosy patients (LL = 30, BL = 30. BT = 30, and TT = 20), 50 tuberculosis cases, and 30 normal controls. These two LATs were compared with corresponding ELISAs (ND-O-BSA ELISA and PGL-I ELISA) and analyzed by the chi-squared test. There were no significant differences between the two LATs (PGL-I LAT and ND-O-BSA LAT) and their corresponding ELISAs. There was an increase in the proportion of positive cases detectable which coincided with the clinical classification of leprosy, i.e., lepromatous cases were more likely to be positive than tuberculoid cases. LATs are more simple and rapid than ELISAs and have high sensitivity (77% in ND-O-BSA LAT, 80.5% in PGL-I LAT) and specificity (99% in both LATs). LATs may become useful tools for the im-munodiagnosis of leprosy in the field. The stability and repeatability of LATs are discussed in detail.RÉSUMÉ
Au cours de cette étude, on a mis au point deux épreuves d'agglutination du latex avec l'antigène phé-noglycolipidique-1 (PGL-1 ), et l'albumine octyl-disac-charidique bovine naturelle du sérum (ND-O-BSA) utilisée comme antigène, chez 110 malades de la lèpre (LL = 30, BL = 30, BT = 30, et TT = 20), chez 50 cas de tuberculose, et chez 30 témoins normaux. Ces deux épreuves d'agglutination du latex ont été comparées avec les épreuves ELISA correspondantes (ND-O-BSA-ELISA et PGL-I-ELISA) et analysées alors par la méthode du chicarré. Aucune différence significative n'a été mise en évidence entre les deux épreuves d'agglutination du latex (PGL-I-LAT et ND-O-BSA-LAT) et les ELISA correspondants. On a toutefois noté une augmentation de la proportion de cas positifs décelables, qui coincidait avec la classification clinique de la maladie, c'est-à-dire que les cas lépromateux avaient davantage de chances de livrer des résultats positifs que les cas tuberculoïdes. Les épreuves au latex sont plus simples et plus rapides que les ELISA, et témoignent à la fois d'une sensibilité élevée (77% pour ND-O-BSA-LAT, 80, 5% pour PGL-I-LAT) et d'une très grande spécificité (99% pour l'une et l'autre des épreuves au latex). Les épreuves d'agglutination au latex pourraient se révéler des instruments utiles dans le diagnostic immunologique de la lèpre sur le terrain. La stabilité et la reproductivité de ces épreuves au latex sont discutées dans le détail.RESUMEN
Se desarrollaron 2 pruebas de aglutinación de látex (LAT) con glicolípido fenólico-1 (PGL-1) y con el di-sacárido natural octil-albúmina sérica bovina (ND-O-BSA) como antígenos, y se aplicaron a 110 pacientes con lepra (LL = 30, BL = 30, BT = 30, y TT = 20), a 50 pacientes con tuberculosis y a 30 controles normales. Estas dos LATs fueron comparadas con los correspondientes enzimoensayos (ND-O-BSA-ELISA y PGL-1-ELISA) y los resultados se analizaron por la prueba de chi-cuadrada. No hubieron diferencias significativas entre las dos LATs y sus correspondientes ELISAs pero hubo un incremento en la proporción de casos positivos détectables, el cual coincidió con la clasificación clínica de la lepra; los casos lepromatosos tuvieron más probabilidad de ser positivos que los tuberculoides. Las pruebas de LATs son más simples y rápidas que las ELISAs y tienen un alto grado de sensibilidad (77% en ND-O-BSA-LAT, 80.5% en PGL-1-LAT) y especificidad (99% en ambas LATs). Las LATs pueden llegar a ser herramientas útiles para el inmunodiagnóstico de la lepra en el campo. La estabilidad y reproducibilidad de las LATs se discuten en detalle.The phenomenon that latex particles may be sensitized by antigenic substances has opened a new field in serologic methodology. Many previous complex procedures have been simplified by the use of sensitized latex particles. Singer and Platz (9) first established the latex fixation test for the serodiagnosis of rheumatoid arthritis. Duboc-zy and White (3) reported use of a latex agglutination test (LAT) for tuberculosis. Recently, Krambovitis, et al. (4) have reported their results for the rapid diagnosis of tuberculous meningitis. The purpose of this paper is to give the preliminary results of a LAT for the rapid diagnosis of leprosy and to compare the LAT with the natural disaccharide-octyl-bovine serum albumin (ND-O-BSA) ELISA (10) and with the phenolic glycolipid-I (PGL-I) ELISA (P. J. Brcnnan, personal communication, 1986).
MATERIALS AND METHODS
Sera. The sera tested were collected from 110 leprosy patients (classified according to the Ridlev-Jopling scale (7) as: LL = 30, BL = 30, BT = 30, and TT = 20), 50 tuberculosis (TB) cases, and 30 normal controls (NC). These sera were stored in a deep freeze (-20ºC) until used.
Antigens. Phenolic glycolipid-I (PGL-I) and the artificial synthetic product ND-O-BSA were kindly provided by Dr. P. J. Brennan (Colorado State University, Fort Collins, Colorado, U.S.A.). They were coated onto 40-well, flat-bottom, polystyrene microtiter plates (Third Factory for Plastic Products, Shanghai, China). The PGL-I suspension was made by Young and Buchanan's method (11). The pure PGL-I in coating buffer (CB) (Na2CO30.398 g, NaHCO3 0.733 g, distilled water 250 ml, pH 9.6) was sonicated for 60 sec with microprobe at 1.5 μm(Ultrasonic Disintegrator; MSE Instrument, England). A milky stable suspension was obtained and then diluted with CB to the required concentration (2.5 μg/ml)for coating the plates. For ND-O-BSA, each vial contained 50 μgof sugar (as glucose equivalent) and 200 μgof BSA and represented 0.5 ml of reconstituted lyophilized material; 0.5 ml of distilled water was added for re-constitution. This solution was diluted with CB to the required coating concentration (0.1 μg/ml) for coating the plates (10). The latex suspension was sensitized with PGL-I and ND-O-BSA as follows: 10% latex suspension was diluted with glycine buffer (GB) (glycine 0.7505 g, 0.1 N NaOH 2.5 ml, distilled water 100 ml, pH 8.2) into a 1% suspension (A); 2 μl of ND-O-BSA solution (200 μg/ml) was added to 1 ml of GB (B); and 50 μlof PGL-I suspension (200 μg/ml) was mixed with 0.95 ml of GB (C). Then, 1 ml of A was added to B. After shaking, the suspension was incubated at 37ºC for 2 hr (shaken again after incubating for 1 hr), and was then ready for use. For PGL-I, the procedures were about the same as those of sensitizing latex particle with ND-O-BSA except that 1 ml of A was added to C.
Blocking agent. Skim milk (SM), an instant nonfat dry milk (Lucerne, U.S.A.), was resuspended with phosphate buffered saline (PBS) (Na2HP04 12.8 g, NaH2PO4 2.62 g, NaCl 0.58 g, distilled water 1000 ml, pH 7.4). Its working concentration was 2.5%.
Conjugates and color reagent. Horseradish peroxidase (HRP) conjugated anti-human IgM (DAKO; Accurate Chemical & Scientific Corp., Westbury, New York, U.S.A.) was diluted with PBS containing 2.5% SM. Its working concentration was 1:2000.
o-Phenylenediamine (OPD) (Sigma Chemical Company, St Louis, Missouri, U.S.A.) was dissolved in citrate buffer (citric acid 4.67 g. Na2HP04 7.3 g, distilled water 1000 ml, pH 5.0) and diluted to the required concentration (0.04%).
Latex agglutination test (LAT). Slides must be very clear and not thick. Before testing, three properly spaced printed circles (1.5 cm diameter) were drawn with nail polish on each slide, and the serum samples were heated at 56ºC for 1 hr to remove nonspecific interference. The tests for detecting antibody were carried out by placing 10 μl of sera onto three circles (the first = normal control, the second = positive control, the third = test serum) of a glass test slide backed with a piece of black paper. Fifteen μl of the lest latex reagent was added to each circle containing the sera, and then the slide was shaken gently until fairly mixed and observed for agglutination. A positive result was indicated by the development of an agglutinated pattern within 3-10 min in the positive control and test serum, with no agglutination in the normal control circle. For a negative result, expressed by the minus sign (-), both normal control and test serum samples showed no agglutination, but the positive control showed an agglutination pattern.
The grade of the agglutination pattern was expressed by the plus sign (+): " + + + " = significant agglutinated rough particles, even agglutination block, and liquid was clear and transparent; " +" = definitely agglutinated minor and homogenous particles, and liquid was clear and transparent; " + +" = between " +" and " + + +."
ELISA. ELISA procedures described by Brcnnan (personal communication, 1986) and Cho, et al. (2)were employed with minor modifications. Briefly, 0.1 ml of PGL-I or ND-O-BSA in CB was added to each well of flat-bottom microliter plates. The antigen-coated wells were blocked to prevent nonspecific binding by adding 0.2 ml of 2.5% SM in PBS (pH 7.4), and incubated at 37ºC for 1 hr. After emptying the blocking agents, 0.1 ml of serum diluted 1:200 in PBS containing 2.5% SM was added to duplicate wells and incubated at 37ºC for 1 hr. After washing four times with PBS, 0.1 ml of per-oxidase-conjugated, anti-human IgM conjugate diluted 1:2000 in PBS containing 2.5% SM was added, and incubated for 1 hr at 37ºC. The plates were washed four times with PBS, and 0.1 ml of the substrate (0.003% hydrogen peroxidase) and the color reagent (0.04% o-phenylenediamine) in citrate buffer were added. The plates were incubated in the dark for 20 min at 37ºC. The reaction was stopped by adding 0.05 ml of 2.5 N sulfuric acid. The results were read at 490 nm with a Model DG-3022 Detector for ELISA (Huadong Electric Tube Factory, Nanjing, China).
Statistical analysis. The upper limits for normal values were calculated by the percentile method (6) in ND-O-BSA ELISA and PGL-I ELISA. The differences between the LATs and their corresponding ELISAs were analyzed by the paired chi-squared test (8). The comparison of two proportions in unpaired cases was conducted using the formula (1) of μ = P1 - 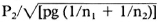 (μ = standardized normal deviate). The relationship between the OD values in the ELISA and grade in the LAT is illustrated with a scatter diagram.
(μ = standardized normal deviate). The relationship between the OD values in the ELISA and grade in the LAT is illustrated with a scatter diagram.
RESULTS
Tests were conducted according to the principle of a double-blind trial. The results of the LATs and ELISAs, using PGL-I and ND-O-BSA as antigens, obtained on the 190 sera shown in Table 1 indicate that: a) with either PGL-I or ND-O-BSA as antigens, the results of the LATs were essentially similar to those of the corresponding ELISA, but the LATs seemed to be more sensitive in BT, especially the PGL-I LAT; b) the results of both the PGL-I LAT and the ND-O-BSA LAT were virtually identical; c) positive cases gradually increased in each method from TT to LL, suggesting a relationship with the bacterial index of the leprosy patients.

Table 2 shows the results of statistical testing of the PGL-I LAT versus the ND-O-BSA LAT, PGL-I LAT versus PGL-I ELISA, and ND-O-BSA LAT versus ND-O-BSA ELISA by paired chi-squared analysis. These results indicate that there were no statistically significant differences for each matched pair.
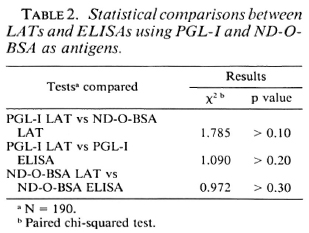
The Figure illustrates the relationship between the LAT grades (LA-G) and the ELISA OD values (EL-OD) in sera from various subjects when PGL-I was used as the representative antigen. The Figure is divided into six small scatter diagrams (SCD) according to the types of sera tested. Analyzing these distributions of EL-OD and LA-G in each small SCD indicated that: a) High EL-OD values were associated with an increase of LA-G and were coincident with the clinical spectrum of leprosy, b) When the EL-OD values were > 0.1, a LAT reaction was sometimes positive in leprosy patients and when the EL-OD was > 0.2, positive cases in LAT significantly increased.
The Figure. Scatter diagrams of LAT grades and ELISA in sera from patients with leprosy (110), tuberculosis (50), and from normal controls (30) with PGL-I as antigen. NC = normal controls; TB = tuberculosis cases; TT, BT, BL, LL = leprosy patients.
Table 3 shows the results of repeated tests for both the PGL-I LAT and ND-O-BSA LAT. By paired chi-squared analysis, there were no significant differences between the first and second tests with the PGL-I LAT or with the ND-O-BSA LAT.

Table 4 shows the results of using a larger sample of sera from patients with leprosy, tuberculosis and from normal controls. From these data: a) The positive rates were 77% (ND-O-BSA LAT) and 80.5% (PGL-I LAT) in the sera of leprosy patients, and were 1% (in both LATs) in TB and NC sera. These results suggest that both LATs were highly sensitive and specific, b) Comparison of two proportions indicate that there were no significant differences between the ND-O-BSA LAT and the PGL-I LAT. c) The positive rates were higher in multibacillary (LL/BL) patients than in paucibacillary (BT/ TT) patients in both the ND-O-BSA LAT and PGL-I LAT.
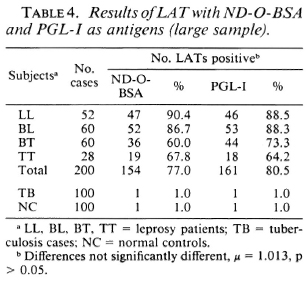
All results of LATs and ELISAs using ND-O-BSA as antigen were similar to those using PGL-I as antigen.
DISCUSSION
Based on polystyrene latex particles sensitized with PGL-I and ND-O-BSA, we developed two kinds of latex agglutination tests-PGL-I LAT and ND-O-BSA LAT. Their sensitivity and specificity were similar to corresponding ELISAs- PGL-I ELISA and ND-O-BSA ELISA. The LATs are more simple and rapid than the ELISAs and do not need special equipment or reagents. With only one drop of scrum, latex, sensitized with the above-mentioned antigens on a slide, can produce the test results. If the LAT kit is stored at 4ºC or with disinfectant (1), its efficiency will be kept for 1 year. Thus, LATs could become more useful tools for the immunodiagnosis of leprosy in the field.
According to our results shown in Table 1, the number of positive cases with LATs using PGL-I and ND-O-BSA as antigens was higher in paucibacillary (BT/TT) leprosy patients than their corresponding ELISAs. From the data in The Figure, it can be seen that in many of the paucibacillary patients' sera low OD cases were negative in the ELISA but positive in the LAT. Thus, the sensitivity of the LAT was higher than that of the ELISA in paucibacillary patients. With TT and BT cases, if the cut-off points in the ELISAs were lowered to OD = 0.15 or OD = 0.10, the positivity rates in the LATs would then more closely correspond to the positivity rates in the ELISAs. These changes in cut-off points would not affect the positivity rates of the LATs or the ELISAs in LL and BL patients' sera. On the other hand, the false-positive rates would be significantly increased in normal and tuberculoid cases if the ELISA cut-off points were lowered. This is a typical consideration of the desired balance between sensitivity and specificity in a serodiagnostic test. In a majority of BT/TT patients' sera, due to their very low content of antibody in sera, the OD values in the ELISA often overlap with those of tuberculosis patients and normal controls. A qualitative test such as the LAT seems promising, and a further comparative study on LATs and ELISAs in a large sample of paucibacillary leprosy patients seems warranted.
For the stability and repeatability of LATs, several technical points such as pH and temperature (12) must be considered, a) Buffer: glycine buffer is better than PBS. the pH of the buffer is one of the most important factors. Lu and Hua (5) reported pH 8.4 is better than pH 8.2, but our LAT results indicated pH 8.2 in our glycine buffer is the best selection, b) NaCl: Lu and Hua (5) reported the optimum concentration for NaCl is 0.3 ~ 1.3% in their test. In our test, if the concentration is more than 0.9% self-agglutination can occur. If no NaCl is used in the latex agent, the agglutinated particles are hazy. c) Washing the latex particles sensitized with testing antigens: Lu and Hua (5) considered it not necessary. Our experience is the same. d) Test sera must be inactivated before testing. Due to the low level of antibodies against Mycobacterium leprae in sera from paucibacillary leprosy patients, at the time of testing the sera should not be diluted; although higher levels of antilepro-sy antibodies are found in sera from multibacillary leprosy patients (scrum dilution may be 1:320 in some individuals), most of them showed best results without dilution or at a 1:2 serum dilution.
REFERENCES
1. Armitrage, P. Statistical Methods in Medical Research. London: Blackwell Scientific Publications, 1983, p. 130.
2. Cho, S.-N.. Yanigahara. D. L., Hunter, S. W., Gelber, R. H. and Brennan, P. J. Serological specificity of phenolic glycolipid I from M. leprae and use in scrodiagnosis of leprosy. Infect. Immun. 41(1983)1077-1083.
3. DuBoczy, B. O. and White, F. C. Latex agglutination test for tuberculosis. Am. Rev. Respir. Dis. 94(1966)914-922.
4. Krambovitis, E., McIllmurray, M. B., Lock, P. E., Hendrickse, W. and Holzel, H. Rapid diagnosis of tuberculous meningitis by latex particle agglutination. Lancet 2(1984)1229-1231.
5. Lu, S.-K and Hua, M.-J. Latex fixation reagent for the detection of rheumatoid factor. Shanghai J. Immunol. 2(1982)24-26.
6. Medical Institute of Si Chuan, China. Medical Statistics 1978. pp. 96-103.
7. Ridley, D. S. and Jopling, W. H. Classification of leprosy according to immunity; a five-group system. Int. J. Lepr. 34(1966)255-273.
8. Shanghai First Medical College. Statistical Methods. Shanghai: Shanghai Science and Technology Publishing House, 1984, pp. 121-122. (x2,fourfold table).
9. Singer, J. M. and Plotz, C. M. Latex fixation test: I. Application to serologic diagnosis of rheumatoid arthritis. Am. J. Med. 21(1956)888-892.
10. Wu., Q.-X., Ye, G.-Y. and Li, X.-Y. Study on serological activity of ND-O-BSA in sera from patients with leprosy, tuberculosis and normal controls. Int. J. Lepr. 56(1988)50-56.
11. Young, D. B. and Buchanan, T. M. A serological test for leprosy with a glycolipid specific for M. leprae. Science 221(1983)1057-1059.
12. Yu, H., Xie, S.-W., Young, G.-Z. and Xu, Y.-P. Latex agglutination reaction. Clin. Immunol. Technol. (1982)93-96.
1. Associate Professor; Institute of Dermatology, Chinese Academy of Medical Sciences, 114 Jiang-wangmiao Road, Taipingmen, Nanjing, People's Republic of China.
2. Professor; Institute of Dermatology, Chinese Academy of Medical Sciences, 114 Jiang-wangmiao Road, Taipingmen, Nanjing, People's Republic of China.
3. Research Assistants; Institute of Dermatology, Chinese Academy of Medical Sciences, 114 Jiang-wangmiao Road, Taipingmen, Nanjing, People's Republic of China.
4. Research Assistants; Institute of Dermatology, Chinese Academy of Medical Sciences, 114 Jiang-wangmiao Road, Taipingmen, Nanjing, People's Republic of China.
5. Research Assistants; Institute of Dermatology, Chinese Academy of Medical Sciences, 114 Jiang-wangmiao Road, Taipingmen, Nanjing, People's Republic of China.
6. Technician, Institute of Dermatology, Chinese Academy of Medical Sciences, 114 Jiang-wangmiao Road, Taipingmen, Nanjing, People's Republic of China.
Reprints requests to Professsor Ye.
Received for publication on 11 July 1988.
Accepted for publication in revised form on 21 November 1989.