- Volume 59 , Number 4
- Page: 618–23
Experimental borderline lepromatous leprosy with intraneural erythema nodosum leprosum in a mangabey monkey (Cercocebus atysy)
ABSTRACT
A sooty mangabey monkey (Cercocebus atys) was inoculated with Mycobacterium leprae and developed borderline lepromatous leprosy and intraneural erythema nodosum leprosum. Previously studied mangabeys have developed only disseminated lepromatous leprosy without reactions. This case broadens the spectrum of leprosy seen in experimentally inoculated animals and further characterizes the nonhuman primate model of leprosy.RÉSUMÉ
On a inoculé un singe mangabey (Cercocebus atys) avec Mycobacterium leprae, et celui-ci a développé une lèpre borderline lépromateuse et un érythème noueux lépreux intraneural. Les singes mangabeys étudiés précédemment avaient seulement développé une lèpre Iépromateuse disséminée sans réaction. Ce cas élargit le spectre de la lèpre observé par l'inoculation expérimentale d'animaux, et fait progresser la caractérisation du modèle primate non humain de la lèpre.RESUMEN
Un mono mangabey pardo (Cercocebus atys) inoculado con Mycobacterium leprae, desarrollo lepra lepromatosa subpolar y eritema nodoso leproso intraneural. Los monos mangabey previamente estudiados solo han desarrollado lepra lepromatosa pero no reacciones leprosas. Este caso amplia la caracterización de este primate no humano como huésped alternativo de la lepra y la información sobre el espectro de la enfermedad en los animales inoculados de manera experimental.Progress in many areas of leprosy research has been impeded by the lack of an animal model that mimics the entire spectrum of the disease, including reactions, that occurs in human beings. Nonhuman primates may offer the best hope for developing such a model. Natural infections in nonhuman primates have been recognized in chimpanzees (5, 11) and in sooty mangabey monkeys (7, 17). During the past century numerous attempts have been made to experimentally infect nonhuman primates with Mycobacterium leprae (16). Successful progressive experimental infection has been reported in a chimpanzee (8), a gibbon (23), and sooty mangabeys, rhesus monkeys, and African green monkeys (25). We have described the postmortem findings in an experimentally infected sooty mangabey (4) and a rhesus monkey (2) with disseminated lepromatous leprosy and in African green monkeys with borderline lepromatous neural leprosy (1). We have also documented the cell-mediated and serologic responses of experimentally infected sooty mangabeys and spontaneously infected chimpanzees (6, 8, 9, 15, 19, 20)
This paper describes the necropsy findings in an experimentally infected mangabey with borderline lepromatous leprosy complicated by intraneural erythema nodosum leprosum.
MATERIALS AND METHODS
A feral born, female sooty mangabey monkey (Cercocebus atys, #D088), acquired as an adult from a commercial vendor, was utilized. She was well nourished, in good health, and tuberculin and simian immunodeficiency virus (SIV) negative. The monkey was caged in an isolation facility at the Tulane Regional Primate Research Center, Covington, Louisiana, U.S.A., fed a commercial diet, and given water ad lib. The mangabey was anesthetized with ketamine HC1 (10 mg/kg) for all procedures requiring handling. She had an infant which was caged with her but which was not on any experimental protocol.
The M. leprae inoculum was prepared from biopsy specimens of cutaneous lepromas taken from an experimentally infected, SIV-positive mangabey (#A022) with disseminated lepromatous leprosy (4). The tissue was homogenized, filtered through gauze, and centrifuged to remove large clumps of debris. Bacilli in the supernatant were counted by the method of Shepard and McRea (22). The inoculum contained 6.9 x 109 acid-fast bacilli (AFB) per ml with 10% solid-staining organisms, hence the inoculum was highly viable. The monkey was inoculated in July 1983 as follows: 3.0 ml intravenously, 1.0 ml flushed intranasally in each nostril, and 0.5 ml intracutaneously at each of eight sites (left forearm, left calf, each ear tip, nose, left eyebrow, lip, and left wrist).
The monkey was lepromin tested 24 months after inoculation using different lepromin preparations as previously described for rhesus monkeys (3).
When the animal died, a complete necropsy was done within 2 hr. Tissues collected for histopathology were fixed in 10% neutral buffered formalin, processed, and embedded in paraffin for routine light microscopy. Sections were cut at 5 and stained with hematoxylin and cosin (H&E), Fite-Faraco acid-fast stain, and by the peroxidasc-antiperoxidase technique for S-100 protein, Leu-7, alpha-1-antitrypsin, and lysozyme.
Serum and smears of nasal mucosa were obtained during periodic clinical examinations. Sera were stored at - 70ºC while nasal smears were stained with the Fite-Faraco stain. Sera were studied by ELISA for the presence of IgG and IgM antibodies to the cell-wall antigen phenolic glycolipid-I (PGLI), an M. leprae-specific epitope, and lipoarabinomannan (LAM), a mycobacterial-common antigen as previously described(6, 9).
RESULTS
One week after inoculation with M. leprae, all of the cutaneous inoculation sites became acutely inflamed. Histologically, these appeared as focal areas of dermal and subcutaneous necrosis and infiltrations of neutrophils intermixed with a few macrophages, lymphocytes, and eosinophils. There were numerous AFB in the infiltrate. These lesions healed, and the monkey never developed any other clinically apparent cutaneous abnormalities. Nasal smears were negative for AFB until 2 days before death.
The animal was lepromin negative (Mitsuda reaction) with all preparations at 24 months postinoculation. There was only slight perivascular cuffing at test sites and bacilli were not well cleared.
The mangabey died of acute gastric distension 32 months after inoculation. At necropsy, the shaved skin was carefully examined and was grossly normal. The stomach was markedly distended with fluid ingesta and gas, and there was a multilobulated leiomyoma in the body of the uterus.
All of the major peripheral nerve trunks were examined and many were abnormal. The right radial nerve was enlarged to a diameter of about 8 mm (normal about 2 mm) where it was located superficially in the forearm. The enlarged segment was nodular and yellow, as were the digital branches. The right ulnar and median nerves were similarly affected, being of normal size while deep to surrounding muscles, and enlarged, nodular, and yellow where superficial. The enlargement became less apparent as the nerves penetrated the wrist and divided into the digital branches. The nerves of the left arm were similar, with a nodular enlargement (3.5 x 0.5 cm) in the distal one third of the left median nerve (Fig. 1). Both axillary nerves and the nerves of the brachial plexus were unremarkable.
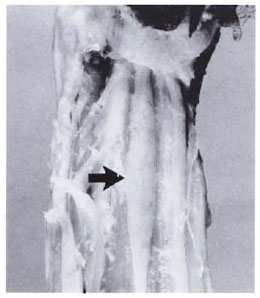
Fig. 1. Enlarged segment of left median nerve (arrow) in a sooty mangabey monkey with borderline lepromatous leprosy.
Within the pelvic limbs, the distal one third of the right tibial nerve was enlarged and brown, gradually increasing in diameter from 2 mm in the normal segment to 5 mm distally. The digital branches were not greatly enlarged, but were discolored. The right superficial peroneal nerve was enlarged to 8 mm in diameter, nodular, and yellow in the distal segment where it emerged to become superficial. The left tibial and superficial peroneal nerves had similar changes. Both sciatic nerves were unremarkable.
Microscopically, there were changes of leprosy in peripheral nerves, skin, peripheral lymph nodes, and the nasal mucosa. The changes in the peripheral nerves varied but, in general, consisted of thickening of the perineurium due to fibrosis and infiltration with lymphocytes, histiocytes, and rare plasma cells. The nerves were slightly to heavily infiltrated with macrophages and lymphocytes, sometimes being completely obliterated by the inflammatory infiltrate (Fig. 2). There were extensive areas of degeneration and disruption of nerve fibers and deposition of necrotic cell debris. In a few areas there was slight endoneural fibrosis. Some macrophages had undergone early epithelioid change, and a few bi- and multinucleated cells were present; however there were no well-developed delayed-type hypersensitivity granulomas. Some macrophages contained globi. Acid-fast staining revealed numerous AFB, singly and in clumps, within perineurial cells, macrophages, and Schwann cells. Most were solidly stained. Stains of infiltrated sections of nerves for S-100 protein revealed virtually no S-100-positivc Schwann cells in the infiltrate, while stains for alpha-1-antitrypsin and lysozyme revealed large numbers of positive monocyte-macrophages.

Fig. 2. Peripheral nerve obliterated by a diffuse inflammatory infiltrate in a sooty mangabey monkey with borderline lepromatous leprosy (H&E x 100).
The right ulnar nerve contained two small (2.5 mm diameter) abscesses composed of neutrophils surrounded by a zone of necrosis (Fig. 3). There were fewer AFB in the abscesses than in other parts of the nerve. These changes are consistent with erythema nodosum leprosum (ENL).
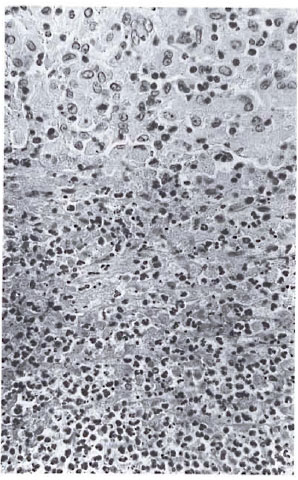
Fig. 3. Abscess typical of ENL in the ulnar nerve of a sooty mangabey monkey with borderline lepromatous leprosy. Note zone of necrosis surrounding neutrophils below (H&E x 250).
There were a few epithelioid cells within the sinusoids of peripheral lymph nodes. These often contained a few AFB. The internal lymph nodes were normal.
The skin of the ear, eyebrow, upper lip, tip of the nose, forearm, wrist, hands, and feet was examined microscopically. In all sections there was minimal-to-mild infiltration of lymphocytes, histiocytes, and rare plasma cells around neurovascular bundles. Many small dermal and subcutaneous nerves were infiltrated with inflammatory cells, and some were fibrotic. Larger subcutaneous nerves had lesions similar to those in nerve trunks. Rare AFB were seen in the dermal infiltrates, while larger numbers were in nerves.
The nasal mucosa and associated nerves were heavily infiltrated with macrophages and lymphocytes. There were numerous well-stained AFB within macrophages.
No inflammatory infiltrates or AFB were seen in the liver, spleen, or bone marrow.
The ELISA anti-PGL-I and anti-LAM showed that prior to M. leprae inoculation (zero time), the animal had an elevated IgG anti-LAM level but negligible IgM anti-LAM and IgG or IgM anti-PGL-I (Fig. 4). Thereafter, within 3 months the IgG anti-PGL-I level rose steeply to >0.3 optical density (OD) and remained elevated, together with IgG anti-LAM, until death. Prior to death, the IgM anti-PGL-I level had also risen to >0.3 OD (Fig. 4). IgM anti-LAM remained relatively low over the course of the study. The curves in Figure 4 have been smoothed to facilitate their visualization.
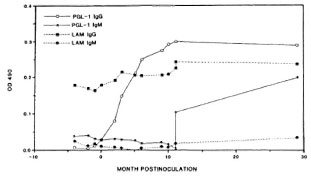
Fig. 4. Levels of anti-PGL-1 and anti-LAM IgG and IgM in the serum of a sooty mangabey monkey inoculated with M. leprae at time 0. Curves have beensmoothed to facilitate interpretation.
DISCUSSION
This case is best classified as borderline lepromatous leprosy. The occurrence of this form of leprosy in an experimentally infected mangabey monkey is important because it broadens the spectrum of leprosy seen in the mangabey. The spontaneously infected mangabeys and previously described experimentally infected mangabeys have had disease at or very near the lepromatous end of the spectrum (4, 17, 25). This is also true for rhesus monkeys, although African green monkeys (Cercopithecus aethiops) also may develop borderline lepromatous leprosy (1). An animal model that develops the complete spectrum of leprosy as seen in humans is important for comparative studies. One reason an animal model of leprosy is so desperately needed is for vaccine development and testing. The fact that some mangabeys can develop some degree of cell-mediated immunity to M. leprae is thus an important observation.
Mangabey #D088 was negative when lepromin tested 24 months postinoculation. We have previously demonstrated that the lepromin test is useful in rhesus monkeys, although a higher concentration of antigen than that used in humans is required (3). The optimal concentration of antigen for use in the mangabey has not been determined. Therefore, a negative lepromin test may indicate either that the animal was anergic at the time tested or that an insufficient concentration of M. leprae was used.
The progressive nonspecific depression of cellular immune function that we have previously described in experimentally inoculated mangabeys with severe lepromatous leprosy (16) was not present in this animal. Lymphocyte blastogenic responses to mitogens and percentages of lymphocytes staining with OKT8 monoclonal antibodies for the suppressor/cytotoxic subset were normal in numerous tests before and after inoculations with M. leprae (Martin and Gormus, unpublished observations).
The longitudinal ELISA antibody results are consistent with our previous observations which suggest that monkeys with more resistant paucibacillary forms of leprosy produce IgG anti-PGL-I antibody (6). IgM anti-PGL-I antibody levels appear to correlate with the less resistant multibacillary forms of leprosy (6). We also previously observed that mangabeys with levels of IgG anti-LAM below 0.5 OD prior to M. leprae inoculation developed more resistant forms of leprosy, consistent with the present observations (9). Mangabeys with preinoculation levels of anti-LAM IgG above 0.5 OD developed multibacillary leprosy (9).
In African green and rhesus monkeys, which arc more resistant than sooty mangabeys to leprosy and which tend to develop ncuritic forms of the disease, we generally observe a longitudinal pattern of elevated IgG and IgM anti-PGL-I coincidentally (Gormus, unpublished observations). Thus, it appears likely that longitudinal monitoring of suspected leprosy patients for ELISA levels of IgG and IgM anti-PGL-I and anti-LAM may be useful in detecting subclinical leprosy, as we have previously suggested (6, 9). It is noteworthy that Levis, el al, have reported increased IgG anti-PGL-I ELISA antibody titers in patients with ENL (14).
The occurrence of ENL in the ulnar nerve of this monkey is also of interest. ENL is one of three reactional states that commonly occur in leprosy patients (l2) and is generally considered to be a form of Arthus reaction (24), although cell-mediated mechanisms may also contribute to the phenomenon(18, 21) . ENL occurs most commonly in lepromatous leprosy, but has been reported in borderline patients (13). The presence of ENL in an animal is important because it expands the spectrum of leprosy lesions seen in nonhuman primates. No animal model has yet manifested the entire range of M. leprae-induced disease that occurs in human beings, but relatively few nonhuman primates have been studied.
The observations reported here further characterize the sooty mangabey monkey as a valuable animal model for leprosy.
Acknowledgment. The authors gratefully acknowledge the helpful comments of Drs. C. K. Job and D. S. Ridley regarding classification of the lesions and the excellent technical assistance of Dorothy Kucbler, Norwood Meincrs, Cyndi Trygg, Susan Coleman, and DorisO'Lcary. Dr. PatrickJ. Brennan, Colorado State University, Fort Collins, Colorado, U.S.A., generously provided PGL-I and LAM antigens for the ELISA test under contract AI-52582. This work was supported by USPHS, NIH grants RR-00164 and AI-19302.
REFERENCES
1. BASKIN, G. B., GORMUS, B. J., MARTIN, L. N., WOLF, R. H., BLANCHARD, J. L., MALATY, R., WALSH, G. P., MEYERS, W. M. and BINFORD, C. H. Experimental leprosy in African green monkeys (Cercopilhecus aethiops): a model for polyneuritic leprosy. Am. J. Trop. Med. Hyg. 37(1987)385-391.
2. BASKIN, G. B., GORMUS, B. J., MARTIN, L. N., WOLF, R. H., MURPHEY-CORB, M., WALSH, G. P., BINFORD, C. H., MEYERS, W. M. and MALATY, R. Experimental leprosy in a rhesus monkey: necropsy findings. Int. J. Lepr. 55(1987)109-115.
3. BASKIN, G. B., GORMUS, B. J., MARTIN, L. N., WOLF, R. H., WATSON, E. A., WALSH, G. P., MEYERS, W. M. and BINFORD, C. H. The lepromin test in rhesus monkeys. Int. J. Lepr. 54(1986)427-436.
4. BASKIN, G. B., WOLF, R. H., GORMUS, G. J., MARTIN, L. N., WALSH, G. P., BINFORD, C. H., MEYERS, W. M. and MALATY, R. Experimental leprosy in the mangabey (Cercocebus atys): necropsy findings. Int. J. Lepr. 53(1985)269-277.
5. DONHAM, K. J. and LEININGER, J. R. Spontaneous leprosy-like disease in a chimpanzee. J. Infect. Dis. 136(1977)132-136.
6. GORMUS, B. J., OHASHI, D. K., OHKAWA, S., WALSH, G . P., MEYERS, W. M., BRENNAN, P. J. and TRYGG, C. Serologic responses to Mycobacterium lepraespecific phenolic glycolipid-I antigen in sooty mangabey monkeys with experimental leprosy. Int. J. Lepr. 56(1988)537-545.
7. GORMUS, B. J., WOLF, R. H., BASKIN, G. B., OHKAWA, S., GERONE, P. J., WALSH, G. P., MEYERS, W. M., BINFORD, C. H. and GREER, W. E. A second sooty mangabey monkey with naturally acquired leprosy: the first reported possible monkey-tomonkey transmission. Int. J. Lepr. 56(1988)61-65.
8. GORMUS, B. J., Xu, K., ALFORD, P. L., LEE, D. R., HUBBARD, G. B., EICHBERG, J. W. and MEYERS, W. M. A serologic study of naturally acquired leprosy in chimpanzees. Int. J. Lepr. 59(1991)450-457.
9. GORMUS, B. J., XU, K., MEYERS, W. M., WALSH, G. P., LEVIS, W . R. and MEEKER, H. C. Antibodies to lipoarabinomannan antigen in sooty mangabey monkeys experimentally inoculated with Mycobacterium leprae. Int. J. Lepr. 58(1990)65-72.
10. GUNDERS, A. E. Progressive experimental infection with Mycobacterium leprae in a chimpanzee. Trop. Med. Hyg. 61(1958)228-230.
11. HUBBARD, G., LEE, D. R. and EICHBERG, J. W. Diseases and pathology of chimpanzees at the Southwest Foundation for Biomedical Research. Am. J. Primatol. (in press).
12. JOLLIFFE, D. S. Leprosy reactional states and their treatment. Br. J. Dermatol. 97(1977)345-352.
13. KARAT, A. B. A., JOB, C. K. and KARAT, S. Erythema nodosum leprosum in borderline leprosy: report of a case. Ing. J. Lepr. 35(1967)17-24.
14. LEVIS, W. R., MEEKER, H. C, SCHULLER-LEVIS, G., SERSEN, E. and SCHWERER, B. IgM and IgG antibodies to phenolic glycolipid I from Mycobacterium leprae in leprosy: insight into patient monitoring, erythema nodosum leprosum, and bacillary persistence. J. Invest. Dermatol. 86(1985)529-534.
15. MARTIN, L. N., GORMUS, B. J., WOLF, R. H., GERONE, P. J., MEYERS, W. M., WALSH, G. P., BINFORD, C. H., HADFIELD, T. L. and SCHLAGEL, C. J. Depression of lymphocyte responses to mitogens in mangabeys with disseminated experimental leprosy. Cell. Immunol. 90(1985)115-130.
16. MARTIN, L. N., GORMUS, B. J., WOLF, R. H., WALSH, G. P., MEYERS, W. M., BINFORD, C. H. and HARBOE, M. Experimental leprosy in non human primates. Adv. Vet. Sci. Comp. Med. 28(1983)201-236.
17. MEYERS, W. M., WALSH, G. P., BROWN, H. L., BINFORD, C. H., IMES, G. D., JR., HADFIELD, T. L., SCHLAGEL, C. J., FUKUNISHI, Y., GERONE, P. J., WOLF, R. H., GORMUS, B. J., MARTIN, L. N., HARBOE, M. and IMAEDA, T. Leprosy in a mangabey monkey-naturally acquired infection. Int. J. Lepr. 53(1985)1-14.
18. MODLIN, R. L., GEBHARD, J. F., TAYLOR, C. R. and REA, T. H. In situ characterization of T lymphocyte subsets in the reactional states of leprosy. Clin. Exp. Immunol. 53(1983)17-24.
19. OHKAWA, S., MARTIN, L. N., FUKUNISHI, Y. and GORMUS, B. J. Regulatory role of FcR+ and FcR-monocyte subsets in Mycobacterium leprae-induced lymphoproliferative response in vitro. Clin. Exp. Immunol. 67(1987)43-50.
20. OHKAWA, S., MARTIN, L. N. and GORMUS, B. J. Lepromin-induced lymphoproliferative response of experimental leprosy monkeys: regulatory role of monocyte and lymphocyte subsets. J. Immunol. 138(1987)3943-3948.
21. RAO, T. D. and RAO, P. R. Enhanced cell-mediated immune responses in erythema nodosum leprosum reactions of leprosy. Int. J. Lepr. 55(1987)36-41.
22. SHEPARD, C. C. and MCRAE, D. H. A method for counting acid-fast bacteria. Int. J. Lepr. 36(1968)78-82.
23. WATERS, M. F. R., BAKRI, I. B., ISA, H. J., REES, R. J. W. and MCDOUGALL, A. C. Experimental lepromatous leprosy in the white-handed gibbon Hylobatus lar: successful inoculation with leprosy bacilli of human origin. Br. J. Exp. Pathol. 59(1978)551-557.
24. WEMAMHU, S. N. C, TURK, J. L., WATERS, M. F. R. and REES, R. J. W. Erythema nodosum leprosum: a clinical manifestation of the Arthus phenomenon. Lancet 2(1969)933-935.
25. WOLF, R. H., GORMUS, B. J., MARTIN, L. N., BASKIN, G. B., WALSH, G. P., MEYERS, W. M. and BINFORD, C. H. Experimental leprosy in three species of monkeys. Science 227(1985)529-531.
1. D.V.M., Tulane Regional Primate Research Center, Tulane University, Covington, Louisiana 70433 and Armed Forces Institute of Pathology, Washington, D.C. 20306-6000.
2. Ph.D., Tulane Regional Primate Research Center, Tulane University, Covington, Louisiana 70433 and Armed Forces Institute of Pathology, Washington, D.C. 20306-6000.
3. M.D., Tulane Regional Primate Research Center, Tulane University, Covington, Louisiana 70433 and Armed Forces Institute of Pathology, Washington, D.C. 20306-6000.
4. Ph.D., Tulane Regional Primate Research Center, Tulane University, Covington, Louisiana 70433 and Armed Forces Institute of Pathology, Washington, D.C. 20306-6000.
5. D.V.M., Tulane Regional Primate Research Center, Tulane University, Covington, Louisiana 70433 and Armed Forces Institute of Pathology, Washington, D.C. 20306-6000.
6. D.V.M., Tulane Regional Primate Research Center, Tulane University, Covington, Louisiana 70433 and Armed Forces Institute of Pathology, Washington, D.C. 20306-6000.
7. M.D., Tulane Regional Primate Research Center, Tulane University, Covington, Louisiana 70433 and Armed Forces Institute of Pathology, Washington, D.C. 20306-6000.
8. Ph.D., Tulane Regional Primate Research Center, Tulane University, Covington, Louisiana 70433 and Armed Forces Institute of Pathology, Washington, D.C. 20306-6000.
9. R. Malaty, M.D., Tulane Regional Primate Research Center, Tulane University, Covington, Louisiana 70433 and Armed Forces Institute of Pathology, Washington, D.C. 20306-6000.
10. M.D., Ph.D., Tulane Regional Primate Research Center, Tulane University, Covington, Louisiana 70433 and Armed Forces Institute of Pathology, Washington, D.C. 20306-6000.
Reprint requests to Dr. Baskin, Pathology Department, Tulane Regional Primate Research Center, 18703 Three Rivers Road, Covington, Louisiana 70433, U.S.A.
Received for publication on 14 November 1990.
Accepted for publication in revised form on 16 July 1991.