- Volume 58 , Number 3
- Page: 466–8
Acid-fast bacilli in semen; correlation with bacterial index
ABSTRACT
Thirty multibacillary patients (12 LL, 18 BL) were historically reviewed and clinically assessed for evidence of testicular involvement. The bacterial index (BI) of the patients ranged f rom 1+ to 6 + ; the morphological index (MI), f rom 0% to 3%. Nine (30%) patients had oligospermia, while acid-fast bacilli (AFB) were demonstrable in the semen of 3 (10%) patients (2 LL, 1 BL). There was a significant correlation between the BI and the demonstration of AFB in semen (p < 0.01).RÉSUMÉ
L'histoire de trente patients multibacillaires (12 LL, 18 BL) fut passée en revue et cliniquement évaluée pour mettre en évidence un envahissement testiculaire. L'indice bactériologique (IB) des patients variait de 1 + à 6 + ; l'indice morphologique (IM) de 0% à 3%. Neuf (30%) patients présentaient une oligospermie, et des bacilles acido-résistants (BAR) pouvaient être mis en évidence dans le liquide séminal de 3 (10%) patients (2 LL, 1 BL). Il y avait une corrélation significative entre l'indice bactériologique et la mise en évidence de bacilles acido-résistants dans le liquide séminal (p < 0.01).RESUMEN
Se revisaron las historias clínicas de 30 pacientes con lepra multibacilar (12 LL, 18 BL) y se estableció el grado de afección testicular. El índice bacteriano (BI) de los pacientes osciló de 1+ a 6+ y el índice morfológico (MI) de 0% a 3%. Nueve pacientes (30%) tuvieron oligospermia y en 3 (10%) (2 LL, 1 BL) se encontraron bacilos acido resistentes (BAR) en el semen. No hubo una correlación significante entre el BI y la demostración de BAR en el semen (p < 0.01).Mitsuda (3) reported that half of the male patients with lepromatous leprosy develop testicular atrophy with direct invasion of the seminiferous tubules and Leydig cells by Mycobacterium leprae. The presence of M. leprae in seminal vesicles is mentioned by Rogers and Muir (5). However, the demonstration of acid-fast bacilli (AFB) in semen has not been commonly described by previous investigators. Skinsnes (7) made a passing mention that AFB may be found in the semen. Pareek and Al-Nozha (4) noted the paucity of literature regarding the demonstration of M. leprae in semen, and reported a 36-year-old male with oligospermia and numerous AFB in the semen. Although the bacilli were not inoculated into mouse foot pads, their morphology resembled that of M. leprae and was unlike M. smegmatis. Singh, et al. (6) more recently demonstrated 1+ to 2+ leprosy bacilli in the semen in two lepromatous patients.
MATERIALS AND METHODS
Thirty male patients clinically diagnosed as lepromatous (LL) or borderline lepromatous (BL) leprosy were selected from the leprosy clinic of the Postgraduate Institute of Medical Education and Research, Chandigarh, India. Untreated patients between the ages of 15 and 50 years were preferred.
A detailed history was taken for each patient which included duration of disease, history of reactions, orchitis, loss of libido, and other factors which could contribute to testicular involvement. A thorough general physical examination and systemic review in addition to a cutaneous examination, slit-skin smears, and skin biopsy were carried out for all of the patients. Slit-skin smears were made from six sites (both earlobes, right elbow, left finger, right knee, and one active lesion).
A testicular examination included the estimation of testicular volume using the Prader orchidometer (normal range = 12 to 25 ml). Testicular consistency, sensation and nodularity of the epididymis were also recorded. Gynecomastia was assessed according to the criterion of a discrete, firm, palpable button of subareolar breast tissue (> 2 cm) with the patient supine.
A sperm count was done following a 5-day period of abstinence. Counts of less than 20 million/ml were considered oligospermia, and this was substantiated by three serial counts done at intervals of 5 days. Azoospermia was the total absence of spermatozoa on all three counts.
For the demonstration of AFB, six thin smears were prepared following liquefaction of the semen sample collected after carefully cleaning the preputial sac. The smears were air dried and stained by the modified Ziehl-Neelsen technique. To retain the morphology of the spermatozoa, the cold method in which the carbol fuchsin was not heated was followed. When AFB were found in the semen, they were confirmed to be M. leprae by loss of acid-fastness with 20% sulfuric acid and lack of growth on Lowenstein-Jensen medium. Mouse foot pad inoculation, however, was not carried out. Renal tuberculosis was ruled out by a 24-hr urine for AFB, a plain x-ray of the abdomen (KUB), and renal function tests.
RESULTS
The 30 patients studied included 12 LL and 18 BL, with a mean age of 32.5 years. The average duration of disease was 2 years. The mean bacterial index (BI) was 2.9 (range = 1 + to 6 + ). The mean Bis in the LL and BL patients were 4.2 and 2.2, respectively.
The morphological index (MI) ranged from 0% to 3%.
Sperm counts revealed 9 patients (30%; 5 LL and 4 BL) having oligospermia, although none had azoospermia. Reduced testicular volume (< 12 ml) could be correlated with oligospermia only in LL patients (p < 0.01); in BL patients this association was not statistically significant (p > 0.05).
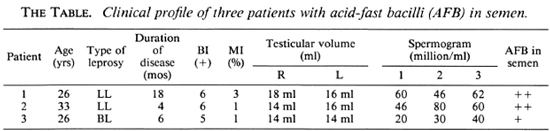
AFB were demonstrable in the semen of 3 patients (10%), 2 of whom were LL and 1 was BL (The Table). The bacilli were present both singly and in groups, and were usually seen as vacuoles in foamy macrophages. Three patients who had AFB in semen had Bis and Mis of 6 + , 3%; 6 + , 1%; and 5 + , 1%. The presence of AFB in semen could not be correlated with duration of disease, past history of reactions, gynecomastia, or testicular atrophy.
DISCUSSION
Although M. leprae have been described in the seminal vesicles (5) and in the seminiferous tubules, they have only rarely been reported in seminal fluid (4,6). The origin of these AFB could be a moot question. They could arise from any of the structures contributing to semen, i.e., testes and epididymis, seminal vesicles, seminiferous tubules, prostate or accessory glands. Further elucidation is required.
The cleaning of the preputial sac and the morphological characteristics preclude any confusion with M. smegmatis (4).
The correlation with a high BI on slit-skin smear has not been attempted by previous authors. This is a significant finding, and can be explained on the basis of a higher bacillemia in these patients (1,2). In addition to being of academic interest, the discharge of bacilli in the semen could be an important mode of transmission of leprosy to a healthy consort directly, and possibly by bed linen and clothes, etc., indirectly.
REFERENCES
1. Chatterjee, G„ Kaur, S., Sharma, V. K., Vaish-navi, C. and Kaur, I. Bacillaemia in leprosy: correlation with slit skin and nasal smears. Indian J. Lepr. 60(1988)535-541.
2. Manja, K. S., Bedi, B. M. S., Kasturi, C, Kirch-heimer, W. F. and Balasuhrahmanyan, M. Demonstration of M. leprae and its viability in peripheral blood of leprosy patients. Lepr. Rev. 43(1972)181-187.
3. Mitsuda, K. Atlas of Leprosy. Tokyo: Chotokai Foundation, 1952.
4. Pareek, S. S. and Al-Nozha, M. Mycobacterium leprae in seminal fluid; a case report. Lepr. Rev. 56(1985)49-50.
5. Rogers, L. and Muir, E. Leprosy. 3rd ed. Bristol: John Wright, 1946.
6. Singh, J., Handa, F., Singh, A., Gupta, S. and Kalla, N. R. Assay of testosterone, FSH and LH and scrum and spermogram in leprosy patients. Indian J. Dermatol. Vencrcol. Lcprol. 54(1988)75-77.
7. Skinsnes, O. K. Immuno-pathology of leprosy: the century in review; pathology, pathogenesis and development of classification. Int. J. Lepr. 41(1973)329-360 (227 refs).
1. M.D., Junior Resident; Department of Dermatology, Postgraduate Institute of Medical Education and Research, Chandigarh 160012, India.
2. M.D., F.A.M.S., Professor and Head; Department of Dermatology, Postgraduate Institute of Medical Education and Research, Chandigarh 160012, India.
3. M.D., Assistant Professor, Department of Dermatology, Postgraduate Institute of Medical Education and Research, Chandigarh 160012, India.
Reprint requests to Professor Kaur.
Received for publication on 18 October 1989.
Accepted for publication on 19 March 1990.