- Volume 57 , Number 3
- Page: 622–7
Short-term multidrug therapy in multibacillary leprosy-review of 80 cases in two provinces of China (1983-1988)
ABSTRACT
Thirty-three active multibacillary patients f rom nine counties of Weifang Prefecture, Shandong Province, and 47 active cases f rom Menla County, Yunnan Province, People's Republic of China, were treated with 24 and 27 months of multidrug therapy (MDT), respectively, in 1983. Clinical assessments, smears, and histopathologic examinations were carried out independently by study teams f rom the Institutes of Dermatology of these two provinces. Reexaminations at 12-14 months and at termination of therapy showed marked improvement, and there was continued improvement at 12-18 and 33 months on follow up. Conversion of the bacterial index to negativity was 0/33, 5/47 for the patients f rom Shandong and Yunnan provinces, respectively, at the end of MDT and 2/33 at 12 months' and 17/ 47 at 18 months' follow up, which increased to 21/33 and 26/44 at 33 months' follow up. Regression of specific infiltration was about 21 %-100% after 24-27 months of MDT; further regression to 95%-100% occurred at 33 months' follow up.RÉSUMÉ
En 1983, on a traité par la polychimiothérapie, pendant respectivement 24 et 27 mois, 33 malades multibacillaires actifs de 9 cantons de la préfecture de Weifang, dans la province du Shandong, et 47 cas actifs du canton de Menla, au Yunnan, en Chine. Des équipes d'évaluation des instituts de dermatologie de ces deux provinces ont mené, de façon indépendante, des études cliniques, bactériologiques et histopathologiques sur ces malades. Des examens pratiqués 12 à 14 mois après le début du traitement, ainsi qu'à la fin de celui-ci, ont révélé des améliorations notables. Les améliorations observées ont continué à être observées après 12 à 18 mois, et après 33 mois de suivi. Un virage de l'index bactériologique de positif à négatif a été constaté après la fin de la polychimiothérapie ecz respectivement 0 des 33 malades de Shandong, et chez 5 des 47 malades du Yunan. La négativité a été obtenue chez 2 des 33 malades du Shandong après 12 mois, et chez 17 des 47 malades du Yunnan après 18 mois de suivi; ces chiffres se sont élevés respectivement à 21 sur 33 et à 26 sur 44 après 33 mois de suivi. Une régression de l'infiltration spécifique allant de 21 à 100% été observée après 24-27 mois de polychimiothérapie; cette régression a atteint 95%-100% après 33 mois de suivi.RESUMEN
En 1983, treinta y tres pacientes con lepra activa multibacilarde 9 regiones de la Prefectura de Weifang, Provincia de Shandong, y 47 casos activos de la region de Menla, Provincia de Yunnan, República Popular China, recibieron un tratamiento con múltiples drogas (TMD) durante 24 y 27 meses, respectivamente. Las evaluaciones clínicas, los estuidos baciloscópicos y los examenes histopatológicos se realizaron independientemente por personal de los Institutos de Dermatología de ambas provincias. Los examenes a los 12-14 meses y a la terminación de la terapia indicaron sorprendentes mejorías y la mejoría continuó a los 12-18 y 33 meses del seguimiento. Al final del TMD, la negativización de los índices bacteriológicos fue de 0/33 y 5/47 para los pacientes de las provincias de Shandong y Yunnan, respectivamente. A los 12 meses de seguimiento, la negativización ocurrió en 2 de los 33 casos en Shandon y en 17 de los 47 casos en Yunnan a los 18 meses. A los 33 meses de seguimiento, la negativización aumentó a 21/33 y 26/44, respectivamente. La regresión de la infiltración específica fue del 21 al 100% después de 24 a 27 meses de TMD, y del 95 al 100% a los 33 meses de seguimiento.At the Second National Leprosy Conference (Guangzhou, 1981), based on her past experience in leprosy control, China proclaimed her intention to eliminate leprosy before the end of this century and, at the same time, changed the national policy of leprosy control from segregation plus dapsone monotherapy to domiciliary treatment with multidrug therapy (MDT). With the aim to enhance this goal and to test the efficacy of short-term MDT as recommended by the World Health Organization (WHO) (8), 33 active multibacillary (MB) patients from nine counties of Weifang Prefecture in Shandong Province and 47 active MB patients from three leprosy settlements of Menla County in the Xishuanbanna Dai Autonomous Region of Yunnan Province were selected and MDT implemented for 24 and 27 months, respectively. This paper presents the findings in these two groups of patients before and during MDT as well as their surveillance for 33 months after MDT.
PATIENTS AND METHODS
Selection of patients and delivery of drugs. Active MB patients without previous therapy or with only irregular dapsone (50-100 mg/day) monotherapy or dapsone combined with rifampin (600 mg 1/month) for less than 6 months were selected by separate teams organized by the Provincial Institutes of Dermatology of these two provinces. Patients with tuberculosis or other systematic diseases or who were pregnant were not included in this study. A complete history was taken and a clinical examination, slit-skin smears for bacterial index (BI) and a biopsy of active lesions for histopathology were made for each patient. The main characteristics of these two groups of patients are given in Table 1.
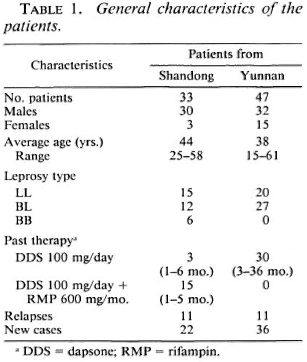
The WHO regimen for MB patients was delivered at monthly intervals by the designated leprosy field worker of the respective County Skin Disease Control Station with supervised intake of rifampin 600 mg, clofazimine 300 mg plus dapsone 100 mg. At each delivery the capsules and tablets were counted to check on the daily doses of clofazimine (50 mg) and dapsone (100 mg) taken during the month. There were 24 total deliveries for the 33 MB patients from Shandong (September 1983-August 1985) and 27 deliveries for the 47 MB patients from Yunnan (February 1983-April 1985). After 24 dosages of MDT, the MB patients from Shandong received 24 additional monthly doses of placebos imitating the three drugs. During each drug delivery, physical changes of the patients' skin and nerve lesions were noted and any signs of drug or leprosy reactions were recorded.
Clinical assessment and laboratory investigations. The team from Shandong consisted of the physician in charge of leprosy from the Provincial Institute of Dermatology, Jinan; the leprosy officer from Weifang Prefectural Bureau of Health plus the team leader of the respective County Skin Disease Control Station. Together they made the pretreatment examinations, visited the patients every 3 months during MDT for clinical assessments, and again every 6 months after MDT. The team from Yunnan was organized in a similar manner. Together with the principal investigator (HYL), they made the clinical and laboratory investigations before treatment and at 14 and 27 months of MDT and at 18 and 33 months after MDT.
The clinical examination included a complete history plus detailed examination for physical changes of leprous lesions as revealed by degrees and areas of erythema, infiltration, papules, nodules, and plaques, as well as areas of anesthesia.
Routine slit-skin smear examinations for acid-fast bacilli (AFB) and bacterial index (BI) were made from one eyebrow, one earlobe, the chin (Shandong), above the knee (Yunnan), plus two lesions marked by the clinical investigator from the local Provincial Institute of Dermatology. Smears were taken by the technician from the respective County Skin Disease Control Station in Wei fang Prefecture (Shandong Province), heat-fixed, and sent to the Provincial Institute of Dermatology in Jinan for staining and reading of the BI. In Menla County (Yunnan Province) all smears obtained before, during, and after MDT were from two technicians (KZ and the principal technician from Xishuanbanna). Staining and reading of the BI were also carried out by them in Menla.
Biopsies were obtained through incision of active lesions chosen on initial examination and immediately adjacent to the original lesions in subsequent biopsies. They were fixed in 4% neutral Formalin solution and dispatched to the respective Institute of Dermatology for processing and readings of the granuloma fraction (GF), bacterial index of the granuloma (BIG), and logarithmic index of biopsies (LIB) as described by Ridley (5).
Clinical assessment and criteria for cure were based on the rules laid down at the National Leprosy Technical Conference in Fuzhou in 1963 and revised in Nanjing in 1982, which are briefly outlined in Table 2.

RESULTS
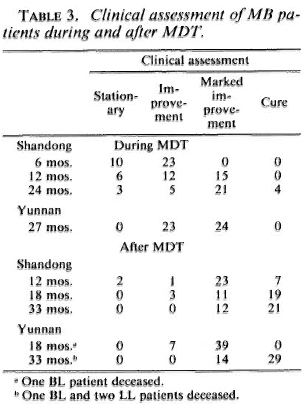
All patients were assessed independently according to the standards given in Table 2 by the study teams of these two provinces. There were 63.6% (21/33) with marked improvement and 12.1% (4/33) with clinical cure in the patients from Shandong and 51.5% (24/47) with marked improvement in the patients from Yunnan at the end of MDT, which increased to 63.6% (21/33) with clinical cure in Shandong and 65.2% (30/44) with clinical cure in Yunnan at 33 months' follow up (Table 3).
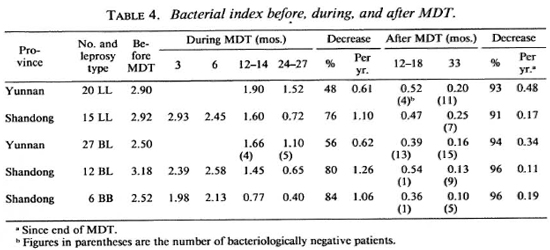
There was a gradual decline in the BI value; 48%-76% for lepromatous (LL), 56%80% for borderline lepromatous (BL), and 84% for mid-borderline (BB) leprosy patients during MDT. The average annual decrease of the BI was 0.61 (LL) to 0.62 (BL) during MDT and 0.34 (BL) to 0.48 (LL) after MDT for the patients from Yunnan. The average annual decreases of the BI for the patients in Shandong were 1.10 (LL) to 1.26 (BL) during MDT and 0.11 (BB) to 0.19 (BL) after MDT (Table 4). This difference is believed due to the variation in the collection of smears by the different technicians from the County Skin Disease Control Stations in Shandong. Conversion to a negative BI was 10.6% (5/47) after 27 months of MDT and increased to 59.1% (26/44) at 33 months' follow up for the patients from Yunnan; the figures from Shandong were 0% and 63.6% (21/33), respectively (Table 4).
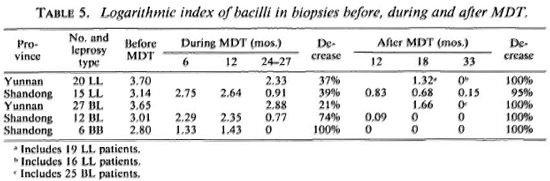
Histopathologic evaluation showed a gradual regression of specific infiltration during MDT, especially for the six BB patients whose infiltrates were reduced to zero at the end of MDT. There was further regression for the rest of the patients after termination of MDT. At the time of the 33 months' follow up, only one LL patient from Shandong showed a decrease of the granuloma fraction (GF) from a pretreatment value of 0.4 to 0.1 at 24 months of MDT, falling to 0.05 at 33 months' follow up. The bacterial index of the granuloma (BIG) remained 3-4+ throughout this period. Altogether there were 20 patients with a GF of 0 and 54 patients with a GF of 0.05-0.10 at 33 months' follow up. These patients, except for the patient mentioned above, were all negative for AFB in the same biopsies. There were a total of 74 biopsies at 33 months' follow up, 4 LL patients from Yunnan died due to other causes after MDT, and 2 BL patients were not available for biopsy (Table 5).
Taken as a whole, 5% (4/80) met the criteria of cure following 24-27 months of MDT, and this increased to 24% (19/79) at 18 months and to 67% (51 /76) at 33 months' follow up after stopping MDT.
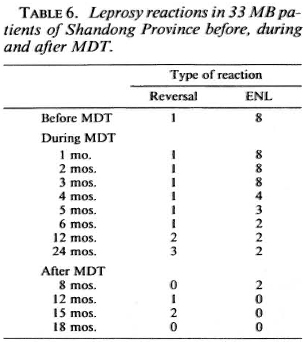
Before MDT, one third of the two groups of MB patients (Shandong 8, Yunnan 14) had frequent erythema nodosum leprosum (ENL) reactions. These ENL reactions subsided gradually within 6-12 months of MDT. Only three patients required increased doses of clofazimine to be weaned from steroids. Two BB patients from Shandong developed reversal reactions at 15-18 months after MDT and were controlled with prednisone 30-40 mg per day (Table 6).
The main side effect was discoloration of the skin, especially in the exposed parts and infiltrated areas; it appeared pink within 1 month of MDT and gradually changed to brownish-red and, finally, to purple (even to violet in some patients). Two patients (M33 and F20), who were at first clinically classified as BT and BB but were LLs histopathologically, did not develop evident pigmentation. At 24-27 months of MDT one patient from each group had regained normal skin color, and the rest of the patients were generally normal within 1 year after MDT was discontinued.
About one third of the patients developed dryness of the skin accompanied by pruritus, especially during the winter months. This dryness of the skin disappeared gradually after cessation of therapy. A few patients complained of anorexia and nausea, which disappeared spontaneously within a few weeks.
Routine blood and urine examinations and liver function tests were performed every 3 months during MDT on the 33 patients from Shandong. The results were all within normal limits.
DISCUSSION
MDT has a very good therapeutic cfTect on lepromatous and borderline patients; the side effects are tolerable and the therapy may be applied in areas without adequate medical facilities. After 24-27 months of MDT, the patients continued to improve clinically, bactcriologically and histopathologically after treatment was discontinued. Thus, the assumption that chemotherapy has to be continued until all criteria of "cure" have been met should be readjusted.
Conversion from smear positive to smear negative has been reported after termination of 12 months of combined therapy which included rifampin (4) and in the follow-up investigations of the Malta Project 3). The THELEP trial with different regimens of MDT indicates that persisters may be detected in all regimens during and at the end of a trial of 24 months (6). In studies of MB patients after prolonged dapsone monotherapy, smears and histology of skin lesions were negative but biopsies of the posterior tibial nerves showed BL-LL histopathology and were positive for AFB. Two BL patients with an additional 3-12 months of MDT had BL histopathology with a 131 of 1-4+ in the tibial nerve (10). On the other hand, it is well known that pure neural leprosy although routinely bactcriologically negative by skin smears may show tuberculoid (2) to even borderline lepromatous histology (7) in the nerve lesions. Thus, it may be concluded that conversion to skin-smear negativity after prolonged chemotherapy is no guarantee for cure, and that persisters also may not be completely eliminated after 24 months of MDT with daily rifampin (6). The different antileprosy drugs in the combined therapy are complementary, designed to be active against each other's drug-resistant mutants and to eliminate those persisters which start to multiply during the course of MDT (7).
The problems of relapse in MB patients after a short course of MDT is not yet well known. Therefore, after 24 months of MDT, although they are clinically cured, it is necessary to follow these patients up for a prolonged period to determine whether the annual relapse rate is within acceptable levels (< 1%) and to determine whether or not the relapse, if it occurs, is with drug-sensitive organisms.
In order to achieve our goal-to eliminate leprosy before the end of the 20th century-it is necessary to detect patients as early as possible and to put them under a regular short course of MDT, and then to apply the manpower thus saved for more health education and case-finding. Our aim is a wide and thorough coverage and proper implementation of MDT to interrupt transmission and to reduce relapses to a minimal level. Our aim is not prolonged MDT to eliminate persisters from individual patients. A survey of 2877 MB patients after clinical cure plus 5 years of maintenance therapy indicated that about 40% (65/163) of relapses occurred between 6-10 years and 18% (30/163) between 11-18 years after clinical cure (9). This delay is perhaps due to maintenance therapy but, until more data are available, it is suggested that after a short course of MDT for MB patients surveillance should be extended to beyond 5 years in carefully selected MDT areas to determine the annual relapse rate due to persisters and their drug sensitivity.
Acknowledgment. We are most grateful to the World Health Organization and the Damien Foundation for the timely and generous gift of clofazimine.
REFERENCES
1. GROSSET, J. H. Recent developments in the field of multidrug therapy and future research in chemotherapy of leprosy. Lepr. Rev. 57 Suppl. 3(1986)223-234.
2. Ji, B. and SHANG, J. [Study of 311 cases of neural leprosy.] Chin. J. Dermatol. 9(1963)18-22.
3. JOPLING, W. H., RIDLEY, M. J., BONNICI, E. and DEPASQUALE, G. A follow-up investigation of the Malta-Project. Lepr. Rev. 55(1984)247-253.
4. PATTYN, S. R., SAINT ANDRÉ, P., FERRACCI, C. and BAQUILLON, G. Comparative study of two regimens of combined chemotherapy of one year duration in multibacillary leprosy. Results after five years' follow-up. Int. J. Lepr. 52(1984)297-303.
5. RIDLEY, D. S. Skin Biopsy in Leprosy. 2nd ed. Basle: CIBA-GEIGY Limited, 1985, p. 59.
6. SUBCOMMITTEE ON CLINICAL TRIALS OF THE CHEMOTHERAPY OF LEPROSY (THELEP) SCIENTIFIC WORKING GROUPOFTHE UNDP/WORLD BANK/WHO SPECIAL PROGRAMME FOR RESEARCH AND TRAINING IN TROPICAL DISEASE. THELEP controlled clinical trials in lepromatous leprosy. Lepr. Rev. 58(1987),325-337.
7. WATERS, M. F. R., RIDLEY, D.S. and RIDLEY, M. J. Clinical problems in the initiation and assessment of multidrug therapy. Lepr. Rev. 57 Suppl. 3(1986)92-100.
8. WHO STUDY GROUP. Chemotherapy of leprosy for control programmes. Geneva: World Health Organization, 1982. Tech. Rep. Ser. 675.
9. ZHENG, D, Study of relapse among 7445 clinically cured leprosy patients in Weifang Prefecture of Shandong Province, China. Abstract in Int. J. Lepr. 57(1989)
10. ZHU, W., ZHAO, B., XIA, M., GU, C, LO, H. and GAO, X. [Study of pathologic changes of posterior tibial nerve in arrested leprosy patients.] J. Clin. Derm. 17(1988)114-119.
1. M.D., M.P.H., Senior Researcher, Beijing Tropical Medicine Research Institute, Beijing.
2. M.D., Deputy Chief; Leprosy Section, Shandong Provincial Institute of Dermatology, Jinan.
3. M.D., Physician, Leprosy Section, Shandong Provincial Institute of Dermatology, Jinan.
4. M.D., Physicianin-Charge of Leprosy, Weifang Prefectural Institute of Dermatology, Weifang, Shandong Province.
5. M.D., Deputy Director; Yunnan Provincial Institute of Dermatology, Kunming.
6. M.D., Physician-in-Charge of Leprosy, Yunnan Provincial Institute of Dermatology, Kunming.
7. B.S., Chief Technician, Wenshan Regional Institute of Dermatology, Wenshan, Zhuang Autonomous Region, Yunnan Province.
8. B.S., Team Leader, Skin Disease Control Station, Menla County, Xishuanbanna Dai Autonomous Region, Yunnan Province, People's Republic of China.
Received for publication on 27 October 1988.
Accepted for publication in revised form on 23 February 1989.