- Volume 57 , Number 3
- Page: 659–70
Intracellular parasitism of parenchymal cells by Mycobacterium leprae1
ABSTRACT
The liver, skeletal muscle, and adrenal gland obtained f rom two nine-banded armadillos infected with Mycobacterium leprae were studied using an electron microscope. M. leprae were found in varying numbers inside hepatocytes, Kupffer's cells, striated muscle cells, adrenal cortical and adrenal medullary cells, endothelial cells, and macrophages. There was evidence to suggest that M. leprae were actively phagocytoscd by the liver and skeletal muscle cells. The inert nature of M. leprae and its behavior as an almost ideal parasite of parenchymal cells are emphasized. The question of whether this unique parasitism of parenchymal cells and the possible processing and presentation of M. leprae antigens by these cells could be responsible for aberrant immune responses is raised.RÉSUMÉ
On a procédé à une étude par microscopie électronique du foie, de muscles striés et des glandes surrénales, sur des échantillons obtenus à partir de tatous à neuf bandes, infectés par Mycobacterium leprae. On a décelé M. leprae en nombre variable à l'intérieur des hepatocytes, des cellules de Kupffer, des cellules de muscle strié, des cellules de la capsule surrénale et de la médullaire de cette glande, des cellules endothéliales, et des macrophages. De plus, certaines observations suggèrent que M. leprae était activement phagocyté par les cellules du foie et par celles des muscles du squelette. On souligne que la nature inerte de M. leprae de même que son comportement, en font un parasite presque idéal des cellules parenchymatcuses. On soulève le problème de savoir si les caractéristiques uniques de parasitisme des cellules parenchymateuses présentées par M. leprae, de même que la présentation des antigènes de M. leprae par ces cellules, pourraient être tenues pour responsables des réponses immunitaires aberrantes notées dans cette maladie.RESUMEN
So hizo un estudio por microscopía electrónica del hígado, del músculo esquelético y de la glándula adrenal de dos armadillos de 9 bandas infectados con Mycobacterium leprae. Se encontraron M. leprae en números variables dentro de los hepatocitos, la células de Kupffer, las células del músculo estriado, las células adrcnalcs corticales y medulares, las células endotcliales, y los macrófagos. Hubieron evidencias que sugirieron que el M. leprae fue activamente fagocitado por las células del hígado y del músculo esquelético. Se enfatiza la naturaleza inerte del M. leprae y su comportamiento como un parásito casi ideal de las células parenquimales. Se plantea la posibilidad de que ésta peculiar parasitación de las células parenquimales y su posible función procesadora y presentadora de los antígenos del M. leprae, pudieran ser responsables de las respuestas inmunes aberrantes encontradas en los pacientes con lepra.Pathogenic viruses and rickettsia are obligate intracellular parasites. They are found within phagocytes and in parenchymal cells of organs wherein they multiply and produce disease. In bacterial infections only the phagocytes, namely, the polymorphonuclear leukocytes and the mononuclear phagocytes, such as the blood monocytes and the resident and elicited macrophages in the tissues, ingest the bacteria. To our knowledge, Mycobacterium leprae is the only known exception to this rule. They are known to invade not only the cells of the phagocytic system but also the parenchymal cells of many organs. The invasion of Schwann cells (18) and the invasion of smooth muscle cells (8,14) are quite well known. In this paper, an electron-microscope study of tissues from the liver, adrenal glands, and the tongue from armadillos infected with M. leprae was conducted to determine the extent of this parenchymal cell invasion by M. leprae. The present knowledge and understanding of this unique behavior of M. leprae is far from adequate. A better understanding of this phenomenon could explain some of the unusual immunological responses of the host to M. leprae.
MATERIALS AND METHODS
Adult nine-banded armadillos (Dasypus novemcinctus) weighing between 3 and 5 kg were captured from the wild. They were screened on admission and once every 3 months for naturally occurring lepromatous disease by examining the skin and subcutaneous tissue for nodules, by taking an ear biopsy, and by examining peripheral blood foracid-fast bacilli (AFB). Aftera minimum period of observation of 6 months to ensure the absence of the wild mycobacterial infection, they were infected intravenously with 4.4 x 108 to 4.6 x 108 M. leprae suspended in 1 ml of saline. The infected animals were evaluated once every 3 months for evidence of disseminated lepromatous leprosy. The infection was considered generalized if in addition to skin and subcutaneous nodules there were AFB in the blood and/or in granulomas in the ear biopsies. When the animals showed signs of terminal disease (weakness, lethargy, loss of appetite and loss of weight), they were sacrificed under anesthesia by exsanguination.
From one animal, pieces of the liver and adrenal gland were removed, and from another a piece of tongue was obtained soon after sacrifice. The tissues were cut into 1-mm cubes and fixed in 5% glutaraldehyde in 0.2 M sodium cacodylatc buffer at pH 7.3 for 2½ hr at 4ºC. They were then washed four times each for 20 min using 0.2 M sucrose solution and postfixed for 1½ hr in 1% osmium tctroxide. The tissues were again washed with buffer four times for 20 min each time, and then dehydrated in a graded series of ethyl alcohol. The procedures for infiltration in Spurrs low viscosity resin (Polysciences, Inc., Warrington, Pennsylvania, U.S.A.) were conducted in vacuum. The tissues were immersed in propylene oxide for 1.5 hr, and then in a thin mixture of propylene oxide and Spurr 3:1 for 1.5 hr, 1: 1 for 0.5 hr, 1:3 for 0.5 hr, and two changes of pure resin for 2 hr first and overnight the second time.
After polymerization, one micron sections were made from at least 50 blocks from each specimen and stained according to the method described by Ludcrschmidt (9). With Luderschmidt's method, the nuclei stained dark blue, the cytoplasm light blue, connective tissue pink and M. leprae deep purple-violet. Sections were examined using the light-microscope for histopathologic changes and for M. leprae. Suitable blocks containing cells with M. leprae were chosen and were trimmed appropriately. Thin sections were made, placed in uncoated grids, then stained with uranyl acetate and lead citrate, and viewed in a Philips EM 410 electron microscope.
RESULTS
Liver. A one micron section stained with Luderschmidt's stain showed liver tissue with prominent Kupffer's cells. There was diffuse infiltration of the sinusoids with macrophages and lymphocytes. In some sections small collections of macrophages forming microgranulomas were seen. M. leprae were present inside macrophages, Kupffer's cells, and hepatocytes. The macrophages and Kupffer's cells contained many more bacilli than did the hepatocytes. An electron-microscopic study confirmed all the above findings. Hepatic cells contained many single M. leprae diffusely scattered in the cytoplasm. Some of them were inside phagosomal vacuoles and many others were not and were found free in the cytoplasm (Fig. 1). Electron-transparent material (ETM) around the bacterial cell membrane was frequently seen. A few organisms showed lysosomal material surrounding them (Fig. 1). Liver cells showing large areas of ETM around bacilli producing early foamy change was an interesting but rare finding (Fig. 2). At the peripheral areas of a few liver cells, there were large, irregular, membrane-bound phagosomal vacuoles containing amorphous material (Fig. 3) which appeared to be formed by the fusion of liver cell processes. In one of the vacuoles a cross section of a bacillus surrounded by similar amorphous material was present (Fig. 3). Multivesicular bodies, lipid droplets, and lipofuscin granules were also seen in the liver cells. The Kupffer's cells had abundant cytoplasm with numerous bacilli, most of them were within phagosomes and some within phagolysosomes. There were numerous dense bodies. Many macrophages in the sinusoids were packed with M. leprae (Fig. 4). The endothelial cells of blood vessels also contained bacilli.
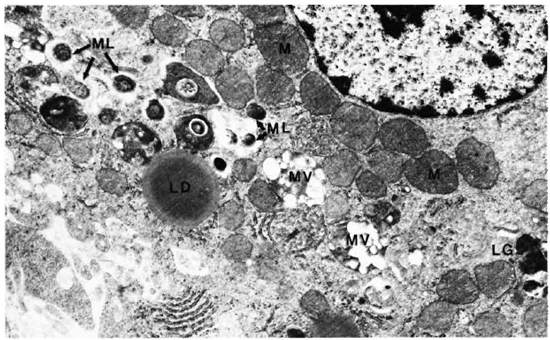
Fig. 1. Hepatic cells with many intracellular M. leprae. M = mitochondria; ML = M. leprae; MV = multivesicular body; LD = lipid droplet; LG = lipofuscin granule (x 12,500).
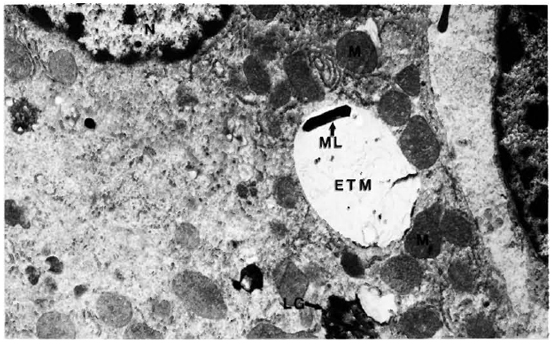
Fig. 2. Hepatic cell with one M. leprae embedded in ETM. M = mitochondria; ML= M. leprae; ETM = electron-transparent material; LG= lipofuscin granule (x 11,500).
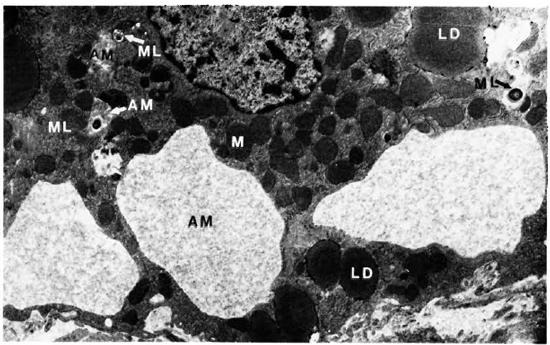
Fig. 3. Hepatic cell with inclusions of amorphous material and M. leprae M = mitochondria; ML = M. leprae; LD = lipid droplet; AM = amorphous material (x 6,800).
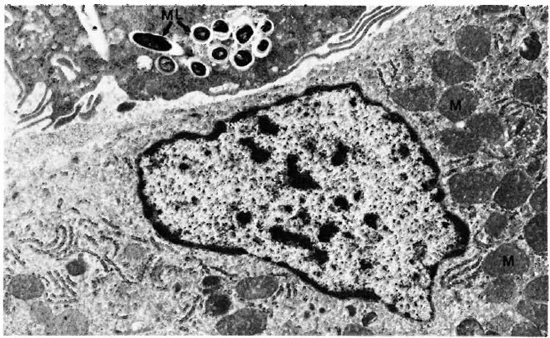
Fig. 4. Hepatic cell with an adjoining Kupffer's cell containing several M. leprae. M = mitochondria; ML = M. leprae (x 11.500).
The majority of the organisms seen in all of these cells were in cross sections. Their cytoplasm was uniformly well stained and their cell membrane was intact. A rare bacillus had a granular and fragmented cytoplasm with collapsed cell membrane.
Adrenal gland. The one micron section showed the adrenal cortex and medulla diffusely infiltrated by scattered macrophages, some of which had a foamy cytoplasm. M. leprae were present in varying numbers inside the macrophages, adrenal cortical cells and adrenal medullary cells. In the cortex, the cells of the zona fasciculata and reticularis had large numbers of bacilli. The zona glomerula showed only scanty organisms. The cells of the adrenal medulla contained clumps of intracellular bacilli.
The electron-microscopic study confirmed these observations. The zona fasciculata of the adrenal cortex was easily identified by the presence of many large and spherical mitochondria with vesicular cristae and light matrix. There were also numerous lipid droplets which, in the deeper parts of the zona fasciculata, were much more osmophilic and tended to be free in the cytoplasm without a limiting membrane (Fig. 5). M. leprae were found singly (Fig. 5) and in clumps (Figs. 6 and 7). It appeared that there was much affinity between the lipid droplets and M. leprae because many of the organisms were found inside the lipid droplets (Fig. 7). Endothelial cells of blood vessels also had many M. leprae.
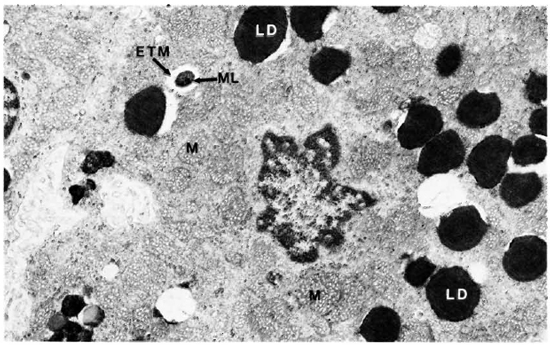
Fig. 5. Adrenal cortical cell with characteristic mitochondria and lipid droplets containing M. leprae. M = mitochondria; LD = lipid droplet; ML = M . leprae; ETM = electron-transparent material (x 11,500).
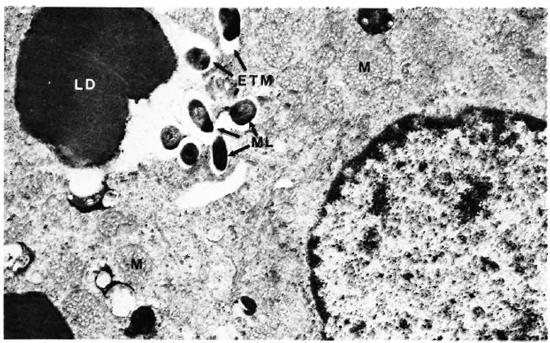
Fig. 6. Adrenal cortical cell with numerous M. leprae. M = mitochondria; LD = lipid droplet; ML = M. leprae (x 16,000).
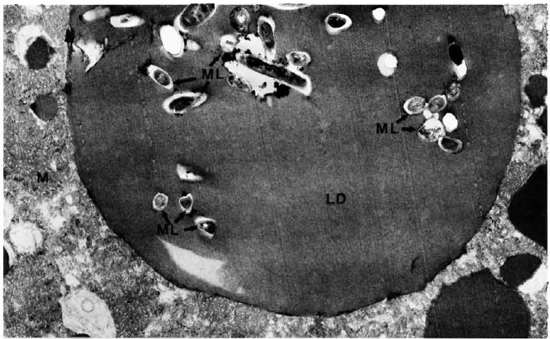
Fig. 7. Adrenal cortical cell with a lipid droplet containing many M. leprae. M = mitochondria; ML = M. leprae: LD= lipid droplet (x 1 1,500).
The organisms found in the cytoplasm were lying free without a phagosomal membrane, and irrespective of their site, whether it was inside the lipid droplet or in the cytoplasm, most of them were surrounded by ETM.
The adrenal medullary cells were recognized by the numerous secreting chromaffin granules. They were osmophilic, and electron dense, membrane bound, round-to-oval in shape, and were distributed throughout the cells. The cells had much smooth endoplasmic reticulum throughout the cell cytoplasm. Mitochondria were small and few (Figs. 8 and 9). M. leprae were present singly (Fig. 8) and in clumps (Fig. 9). Most of the organisms were embedded in ETM (Fig. 9). Where there were many bacteria, many myelin figures were seen (Fig. 9). Macrophages infiltrating the adrenal tissue also contained numerous bacilli (Fig. 10), and were seen in close association with the parenchymal cells. They were very active with many microvilli, mitochondria, and myelin figures. Bacilli were also present inside the endothelial cells of blood vessels.
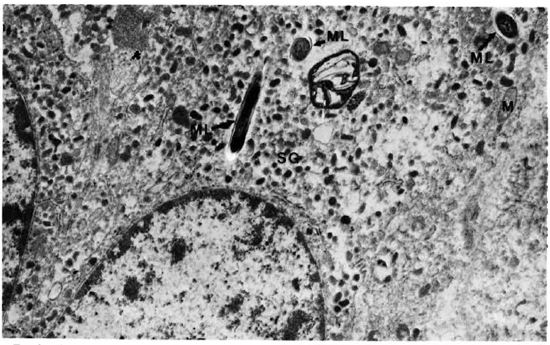
Fig. 8. Adrenal medullary cell with numerous secretory granules and a few M. leprae. ML = M. leprae; M = mitochondria; SG = secretory granules (x 20,500).
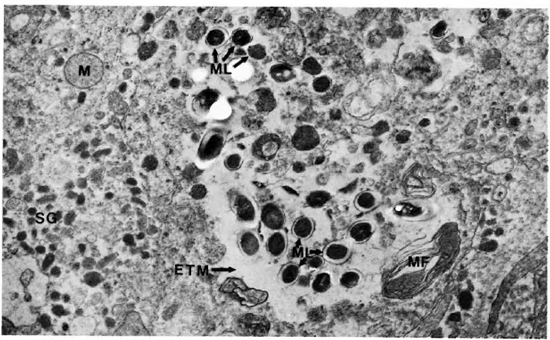
Fig. 9. Adrenal medullary cells with numerous M. leprae with no phagosomal membrane. ML = M. leprae; M = mitochondria; MF = myelin figure; SG = secretory granules (x 20,500).
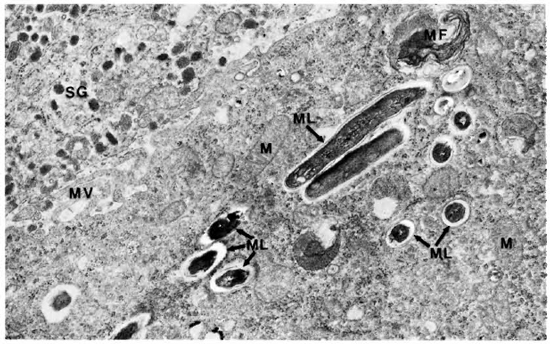
Fig. 10. Adrenal medulla infiltrated by a macrophage with numerous M. leprae. SG = secretory granules; ML = M. leprae; MV = microvilli; MF = myelin figure; M = mitochondria (x 20,500).
Skeletal muscle. The one micron sections of the blocks showed macrophages infiltrating the perimysium and extending along the interstitial space separating muscle fasciculi. Some muscle fasciculi were infiltrated by macrophages and partly destroyed. M. leprae were present inside macrophages, satellite cells, and muscle cells.
The electron-microscopic study showedclearly that macrophages infiltrate and destroy the perimysium and then invade the muscle cells. Their cell borders showed numerous villi, inside some of which were M. leprae (Fig. 11). M. leprae were demonstrated in clumps beyond any doubt inside muscle cells (Fig. 12). The myofibrils appear "to dissolve" away rather than being pushed out. The bacteria were surrounded by increasing amounts of ETM mixed with cell debris. Satellite cells also contained many bacteria (Fig. 13). M. leprae were present in Schwann cells of the nerve, within interstitial tissue, and in endothelial cells.
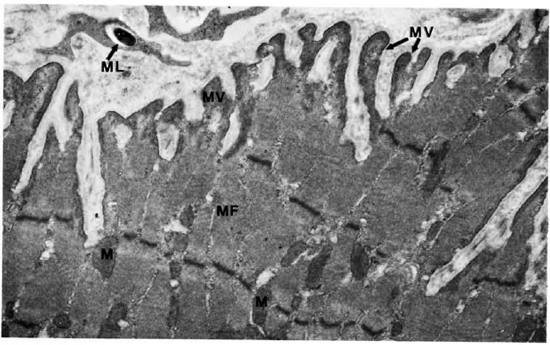
Fig. 11. Skeletal muscle cell with microvilli and a phagocytosed M. leprae. M = mitochondria, MF = myofibril; MV = microvilli; ML = M . leprae (x 13,500).
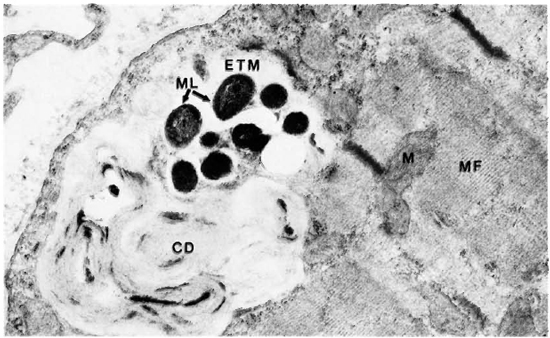
Fig. 12. Skeletal muscle cells with a clump of M. leprae embedded in ETM and cell debris. ML = AL leprae; ETM = electron-transparent material; CD = cell debris; MF = myofibril; M = mitochondria ( x 23,000).
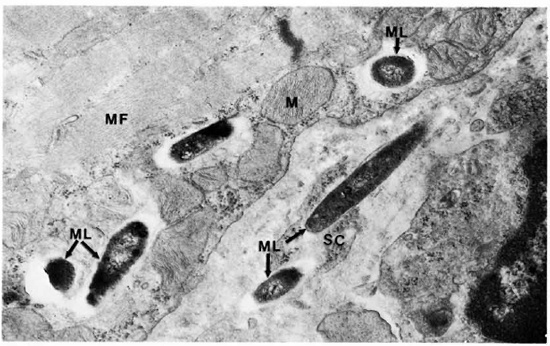
Fig. 13. Skeletal muscle cell and satellite cell containing M. leprae, MF = myofibril; SC = satellite cells; ML = M. leprae, M = mitochondria (30,500).
DISCUSSION
In this electron-microscopic study of experimentally infected armadillo tissues, M. leprae have been conclusively demonstrated in the parenchymal cells of the liver, adrenal gland, skeletal muscle, and endothelial cells. The Kupffer's cells in the liver, the Schwann cells of nerves in the skeletal muscle, and the macrophages infiltrating all of these organs also had intracellular bacilli. Bacilli were found inside phagosomes and also free in the cytoplasm. The characteristic ETM was also present around the bacteria, irrespective of the type of the host cell. All of these findings describing the interaction between M. leprae and its host cells are in agreement with earlier electron-microscopic studies of lepromatous lesions (4,5,12).
The presence of M. leprae in nonphagocytic cells is a feature found in both experimental and human leprosy. In human disease, M. leprae's ability to colonize Schwann cells is one of the essential features searched for to confirm the diagnosis of leprosy. In the earliest skin lesions of leprosy, smooth muscle cells of the arrector pili and Schwann cells of dermal nerves are the main host cells of M. leprae (M). In lepromatous patients, bacilli have been shown inside smooth muscle cells of the dartos tunica (8), inside striated muscle cells (8), cartilage cells (7), and spermatogonia (6). The endothelial cells of blood vessels in many organs, such as the kidney, heart, lungs, and intestinal mucosa, contain M. leprae (2). Recently, it has been stated that endothelial cells could even be preferential sites for the habitat of M. leprae (11).
In experimentally infected nude mice, M. leprae were found in large numbers in striated muscle cells, cardiac muscle cells, cartilage cells, and liver cells (1). In normal mice experimentally inoculated with M. leprae, the organisms are found to multiply in the skeletal muscles of the foot pad (13).
Macrophages are professional phagocytes and phagocytosis of pathogenic bacteria is one of their normal functions. But skeletal and smooth muscle cells, Schwann cells, endothelial cells, and parenchymal cells of the liver and the adrenal gland are not ordinarily phagocytic. For these cells, phagocytosis is an abnormal activity and the presence of large numbers of M. leprae in their cytoplasm needs to be explained.
Phagocytosis can be either nonspecific, as in the nonchordate animal world, or it can be a specific system. Where it involves a specific system, there is first an interaction between the foreign particles and the plasma membrane of the host cell, resulting in attachment of the foreign particle to the cell. Specific recognition is ordinarily mediated by the immune processes. The plasma membrane of macrophages, for example, has Fc and C3b receptors, and when a specific antibody coats a particle and fixes complement, the particle acquires free Fc and C3b regions which can attach to the receptors in the host-cell plasma membrane. The attachment is quickly followed by ingestion, which is described as a zippering process (15). Phagocytosis can also be a nonspecific process as in the case of carbon particles, or physically and chemically altered red blood cells, or poorly virulent and nonvirulent bacteria phagocytosed by macrophages.
In this study, it is shown that the liver cells and skeletal muscle cells have numerous microvilli which apparently have surrounded and engulfed M. leprae. In Figure 3, liver cells are shown with their cytoplasm encircling areas of amorphous granular debris and a cross section of M. leprae is seen along with this granular material. It is reasonable to infer that the M. leprae are engulfed by the cytoplasmic processes. A similar deduction could be arrived at in our examination of skeletal muscle cells (Fig. 11). However, Fc receptors, C3b receptors, cytophilic antibodies, etc., are not known to be present in or associated with parenchymal cells. The observed phagocytosis of M. leprae by these cells should, therefore, only be a nonspecific process.
No bacteria other than M. leprae have been demonstrated inside parenchymal cells of immunologically intact animals to our knowledge. Most of the pathogenic bacteria are too toxic to be carried inside any cell except in phagocytes which are equipped to engulf and, if possible, destroy them. Even bacteria like M. tuberculosis, which do not produce any known toxins and would multiply freely inside phagocytes, are not found in parenchymal cells of immunologically intact persons. Nonpathogenic bacteria are unable to gain entry into the body and establish a foothold unless the host is immunologically compromised. Like many pathogens M. leprae gains entry into phagocytes. In addition, unlike other pathogenic bacteria, it also invades parenchymal cells. This process does not appear to be immunologically mediated, and needs further study.
One of the functions of phagocytic cells is the processing of antigens for presentation to lymphocytes. Macrophages process and present antigens to T cells in association with molecules encoded by class II genes of the major histocompatibility complex (MHC) antigens for the initiation of the cell-mediated immune process (19). In the earliest infections with M. leprae the organisms are phagocytosed by tissue histiocytes and macrophages in the skin. In addition, they invade Schwann cells and smooth muscle cells in the dermis. Schwann cells may have an important role in processing and presenting antigens of M. leprae (10). Recent experiments have shown that murine Schwann cells can present M. leprae antigens in association with class I MHC gene products (16). A similar reaction occurring in human subjects is distinctly possible. The view that antigens can only be processed by a small subset of class II-bearing bone-marrow-derived cells is currently in dispute, and it is suggested that antigen processing can be a function of virtually all cells (3). It is worth investigating if presentation of M. leprae antigens in association with MHC class I antigens by parenchymal cells like Schwann cells could lead to aberrant immune responses.
Persisting organisms sensitive to dapsone even after long periods of dapsone antileprosy therapy are well known. They were isolated from striated muscles, smooth muscles of the dartos tunica, and from peripheral nerves (17). M. leprae continue to remain in muscle cells and Schwann cells of patients after long periods of antileprosy therapy. Parenchymal cells do harbor M. leprae, and they could serve as protected sites for the organism. In parenchymal cells, M. leprae may enjoy relative seclusion from antileprosy drugs by virtue of either reduced metabolic activity, and hence reduced susceptibility, or poor pharmacokinetics. In parenchymal cells, the organism should be less susceptible to conventional cell-mediated responses.
It is interesting to see large numbers of M. leprae inside specialized cells such as those of the adrenal cortex and adrenal medulla with only displacement of the intracellular organelles. There was minimal structural and possibly negligible functional damage to the invaded cells until extensive replacement of the cytoplasm had occurred. Our data suggest that M. leprae is an inert organism and an almost ideal parasite whose survival depends on the sharing of its metabolic activity with the cell without damaging it until its numbers become too large for the cell to support their presence and growth.
Acknowledgments. We are grateful to Mrs. Mary M. Jackson for secretarial assistance. This investigation received financial support from the UNDP/World Bank/WHO Special Programme for Research and Training in Tropical Diseases, Geneva, Switzerland, and Intra-Agency Agreement YO1-AI-50004, National Institute of Allergy and Infectious Diseases, Bethcsda, Maryland, U.S.A.
REFERENCES
1. CHEHL, S., RUBY, J., JOB, C. K. and HASTINGS, R. C. The growth of Mycobacterium leprae in nude mice. Lepr. Rev. 54(1983)283-304.
2. DESIKAN, K. V. and JOB, C. K. Visceral lesions caused by M. leprae; a histopathological study. Indian J. Pathol. Bacteriol. 13(1970)100-108.
3. GERMAIN, R. N. The ins and outs of antigen processing and presentation. Nature 322(1986)687-689.
4. IMAEDA, T. Electron microscopic analysis of the components of lepra cells. Int. J. Lepr. 28(1960)22-37.
5. IMAEDA, T. and CONVIT, J. Electron microscope study of Mycobacterium leprae and its environment in a vesicular lepromatous lesion. Lepr. India 34(1962)267-278.
6. JOB, C. K. Gynecomastia and leprous orchitis; a preliminary study. Int. J. Lepr. 29(1961)423-142.
7. JOB, C. K., KARAT, A. B. A. and KARAT, S. Histopathological appearance of leprous rhinitis and pathogenesis of septal perforation in leprosy. J. Laryng. 80(1966)718-732.
8. JOB, C. K., KARAT, A. B. A., KARAT, S. and MATHAN, M. Leprous myositis-a histopathological and electron-microscopic study. Lepr. Rev. 40(1969)9-16.
9. LUDERSCHMIDT, C. Orientation staining for demonstration of Mycobacterium leprae in semithin sections. Int. J. Lepr. 55(1987)83-87.
10. MSHANA, R. N., HUMBER, D. P., HARBOE, M. and BELEHU, A. Demonstration of mycobacterial antigens in nerve biopsies from leprosy patients using peroxidase-antipcroxidasc immunoenzyme technique. Clin. Immunol. Immunopathol. 29(1983)359-368.
11. MUKHERJEE, A. and MEYERS, W. M. Endothelial cell bacillation in lepromatous leprosy: a case report. Lepr. Rev. 58(1987)419-124.
12. NISHIURA, M. Electron microscopic basis of the pathology of leprosy. Int. J. Lepr. 28(1960)357-400.
13. Palmer, E., Rees , R. J. W. and WEDDELL, A. G. M. Site of multiplication of human leprosy bacilli inoculated into the foot-pads of mice. Nature (Lond.) 206(1965)521-522.
14. RIDLEY, D . S. Pathogenesis of early skin lesions in leprosy. J. Pathol. 111(1973)191-206.
15. SILVERSTEIN, S. C, STEINMAN, R. M. and COHN, Z. A. Endocytosis. Annu. Rev. Biochem. 46(1977)669-722 (315 refs.)
16. STEINHOFF, U. and KAUFMANN, S. H. E. Specific lysis by CD8+ T cells of Schwann cells expressing Mycobacterium leprae antigens. Eur. J. Immunol. 18(1988) 969-972.
17. WATERS, M. F. R., REES, R. J. W., MCDOUGALL, A. C. and WEDOELL, A. G. M. Ten years of dapsone in lepromatous leprosy: clinical, bacteriological and histological assessment in the finding of viable leprosy bacilli. Lepr. Rev. 45(1974)288-298.
18. WEDDELL, G., JAMISON, D. and PALMER, E. Recent investigations into the sensory and neurohistolog ical changes in leprosy. In: Leprosy in Theory and Practice. Cochrane, R. G., ed. Bristol: John Wright & Son, 1959, pp. 96-113.
19. ZINKERNAGEL, R. M. and DOHERTY, P. C. Restriction of in vitro T cell-mediated cytoxicity in lymphatic choriomeningitis within a syngeneic or semiallogeneic system. Nature 248(1974)701-702.
1. M.D., F.R.C.Path., F.A.M.S., Chief; Laboratory Research Branch, GWL Hansen's Disease Center, Carville, Louisiana 70721, U.S.A.
2. B.S., Biological Laboratory Technician, Pathology Research Department; Laboratory Research Branch, GWL Hansen's Disease Center, Carville, Louisiana 70721, U.S.A.
3. M.D., Ph.D., Chief, Laboratory Research Branch, GWL Hansen's Disease Center, Carville, Louisiana 70721, U.S.A.
Received for publication on 21 December 1988.
Accepted for publication in revised form on 28 February 1989.
1. Wayne M. Meyers, M.D., Ph.D., kindly served as Editor in regard to the submission, review, revision, and acceptance of this manuscript.