- Volume 67 , Number 2
- Page: 165–7
"Large numbers" of Mycobacterium leprae are discharged f rom the intact skin of lepromatous patients; a preliminary report<
To the Editor:
In 1970 Pedley in an innovative study of the skin of leprosy patients concluded that the number of Mycobacterium leprae discharged from the intact skin of lepromatous patients was negligible and, therefore, nasal secretions were the major source of infection (6). Although his findings seem to be widely accepted and quoted, no attempt has been made to confirm or disprove his hypothesis. In the method employed in his study, called composite skin contact smears (CSCS), a glass slide was pressed 10 times repeatedly on the skin lesions of lepromatous patients with a high bacterial index (BI), heat fixed, stained for M. leprae , and examined under a microscope with an oil immersion lens. From 11 patients, 34 CSCS were studied and only 20 acid-fast organisms (AFB) were detected. On the basis of this study he concluded "that very few bacilli, if any, are discharged from the intact skin of a lepromatous patient."
In this study under report, biopsies from 13 patients belonging to the lepromatous group with a BI of 4+ and above were randomly chosen from the biopsy files of St. Thomas Hospital and Leprosy Centre, Chettupattu, India, from 1992 to 1998. Fresh sections were cut from the stored paraffin blocks and were stained for AFB using a modified Fite's method (4). AFB were looked for under an oil immersion lens in the epithelial and keratin layers of the epidermis. Whenever AFB were found, their locations were carefully studied. In one newly diagnosed, untreated leproma- tous patient, a modified CSCS method was employed to obtain smears. A glass slide was coated with a thin film of water-soluble glue in an area covering 2 square centimeters. The glue-coated area was pressed against the selected skin lesion 10 times, heat fixed, stained for AFB, and examined under an oil immersion lens. Modified CSCS were obtained from eight sites.
Of the 13 biopsies studied, in 5 there were clumps of AFB in one or more focal areas of the keratin layer of the epidermis numbering over 10 per oil immersion field (Figs. 1 and 2), in 4 there were less than 10 per field, and in 4 no bacilli were detected. In four biopsies there were intracellular collections of AFB in the cells of the basal layer and of the squamous cell layer (Fig. 3). On careful study it was found that AFB were mostly found around the pores in the skin, especially at the exit of the hair follicles (Fig. 4). there were also many bacilli in the cells of the hair follicles and around the hair shaft. In the one untreated lepromatous patient in whom the skin was studied using the modified CSCS technique, large numbers of AFB were found as detailed below:
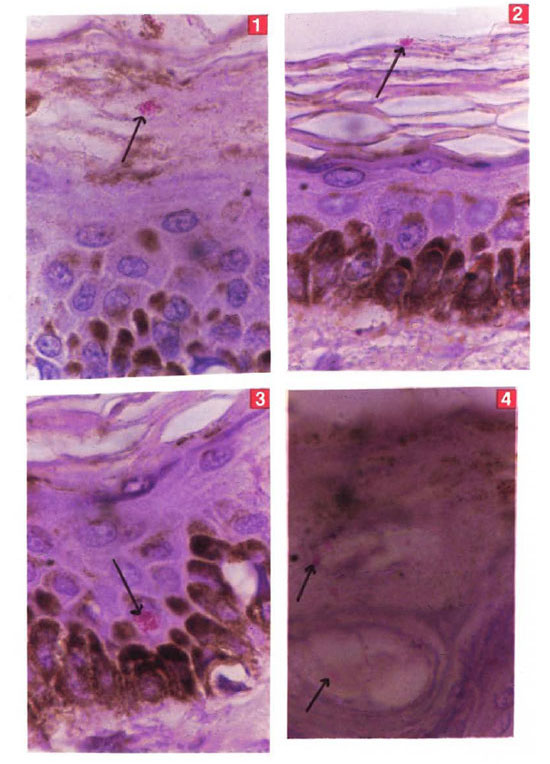
Figs. 1-4. Photomicrographs showing clunnps of. AFB (→) in the keratin layer (Figs. 1 and 2), in the epithelial layer (Fig. 3), and at the exit of a hair follicle (Fig. 4) (H&E x800).
| Right earlobe | Neg. | Right forehead | 13 |
| Left earlobe | 85 | Left forehead | 39 |
| Right upper arm | 24 | Left loin | 35 |
| Left upper arm | 79 | Left thorax | Neg. |
| Total | 275 |
Since the glue used interfered with the staining of AFB, such studies were not done in more patients. Further investigations are being planned to refine the method of detecting AFB in the superficial part of the skin.
In this study it is clearly shown that AFB were present in fairly large numbers in the keratin layer of the epidermis of 9 out of 13 lepromatous patients. AFB were also found in the epithelial cells of the epidermis. In one newly diagnosed lepromatous patient numerous AFB were demonstrated on the surface of the skin using a modified CSCS technique. In 26 to 42 days epidermal cells can travel from the basal layer to the horny layer and, in due course, they are keratinized (3). Obviously, AFB would come up to the keratin layer travelling inside epithelial cells and would be shed into the environment along with keratinized cells. It was also observed that the presence of AFB was concentrated around the pores of the skin, especially those of the hair follicles. It is apparent that the organisms would exit along with the sebaceous secretions. It seems that the method used earlier by Ped- ley (6) (pressing a glass slide against the skin) was not successful in taking up the bacilli which were present in large numbers in the superficial keratin layer of the skin.
In 1964 the majority of leprologists believed that the portal of entry of M. leprae is through the skin, although some believed that the organism entered through the upper respiratory tract (1).It was also thought that direct skin-to-skin contact is likely to be more effective for transmission of the disease than indirect contact (5). Transmission through the respiratory route gained importance following Pedley's conclusion that hardly any M. leprae were discharged into the environment through the skin (6) and the demonstration of enormous numbers of AFB from nasal secretions of lepromatous patients (2).
In conclusion, this study shows that M. leprae are found in the superficial keratin layer of the skin of lepromatous leprosy patients and are shed into the environment in large numbers, thus challenging Pedley's findings. Direct skin-to-skin contact with such patients is more likely to transfer viable M. leprae from a patient to a contact than by any other method. An extensive study is being planned and its results will be reported in due course.
- Charles K. Job, M.D., F.R.C.Path.
J. Jayakumar, M.B.B.S., D.T.M. & H.
M. Aschhoff, M.D.
St. Thomas Hospital & Leproso Centre
Chettupattu 606 801
Tantil Nadu, South India
Acknowledgment. We are grateful to Mrs. K. Jayanthi for secretarial help and to Mr. T. Peter Sathesh and Mr. B. Vijayakumar for technical assistance. The study was financed by the American Leprosy Mission International, Greenville, South Carolina. U.S.A
REFERENCES
1. Badger, L. F. Epidemiology. Chapter VI. In: Leprosy in Theory and Practice . Cochrane R. G. and Davey T. F., eds. Baltimore: The Williams and Wilkins Company, 1964, p. 75.
2. Davey, T. F. and Rees. R. J. W. The nasal discharge in leprosy-clinical and bacteriological aspects. Lepr. Rev. 45(1975)121-124.
3. Halpin, K. M. Epidermal turn over line-a reexamination. Br. J. Dermatol. 86(1972)14-19.
4. Jon, C. K. and Chacko, C. J. G. A modification of File's stain for demonstration of M. leprae in tissue sections. Indian J. Lepr. 58(1986)17-18.
5. Noordeen, S. K. Transmission factor. Chapter 3. In: Leprosy. Vol. II . Dharmendra, ed., Bombay: Samant and Company, 1985, pp. 1097-1103.
6. Pedley, J. C. Composite skin contact smears: a method of demonstrating the nonemergence of Mycobacterium leprae from intact lepromatous skin. Lepr. Rev. 41(1970)31-43.