- Volume 67 , Number 3
- Page: 243–9
Seroprevalence rates of antibodies to phenolic glycolipid-I among school children as an indicator of leprosy endemicity
ABSTRACT
In order to study whether the seroprevalence of antibodies to phenolic glycolipid-I (PGL-I) among school children is a useful indicator of the leprosy problem in certain areas, school surveys were carried out. These surveys have the advantage of targeting an easily accessible, stable and standardized population. Antibodies to the species-specific PGL-I of Mycobacterium leprae were detected in a simple gelatin particle agglutination test. We have determined the seroprevalence rates in 2835 school children f rom five different areas in three provinces of Sulawesi, Indonesia. Three areas with a case-detection rate of over 3.4/10,000 were designated as high-endemic areas. The other two were designated as low-endemic areas, having a case-detection rate of less than 1/10,000. The seroprevalence rates in the three high-endemic areas ranged f rom 26% to 28% (95% CI 21 %-31 %). In both low- endemic areas the seroprevalence rate was 7% (95% CI 5%-10%). In a second survey conducted in one high-endemic area 3 years after the first survey, the seroprevalence rate was the same as in the first survey. These results indicate that seropositivity rates among school children may reflect the leprosy incidence. They illustrate the potential applicability of seroprevalence as an indicator of the magnitude of the leprosy problem in a selected area.RÉSUMÉ
Afin de savoir si la séro-prévalence des anticorps dirigés contre le glycolipide phénolique-I (PGL-I) parmi les enfants scolarisés est un indicateur utile de tendance vers un problème de lèpre dans certaines zones, des enquêtes dans les écoles furent réalisées. Ces enquêtes ont l'avantage de cibler une population facilement accessible, relativement stable et bien standardisée. Les anticorps spécifiques d'espèce, dirigés contre le PGL-I de Mycobacteríum leprae, furent détectés un utilisant un test simple d'agglutination sur particule de gélatine. Nous avons obtenu les taux de séro-prévalence chez 2835 enfants allant à l'école dans 5 zones de 3 provinces du Sulawesi en Indonésie. Trois zones où le taux de détection de cas est au- dessus de 3,4 pour 10 000 furent désignées zones hautement endémiques. Les 2 autres furent qualifiées de zones de faible endémicité, ayant un taux de détection de cas de moins de 1 pour 10 000. Les taux de séro-prévalence dans les 3 zones variaient de 26% à 28% [Intervalle de confiance à 95% (IC95%) 21%- 31%]. Dans les 2 zones à faible endémicité, le taux de séro-prévalence était de 7% (IC95%: 5%-10%). Au cours d'une seconde enquête menée dans une zone hautement endémique 3 années après la première, le taux de séro-prévalence était identique à la première. Ces résultats suggèrent que le taux de séro-prévalence parmi les enfants scolarisés reflète l'incidence de la lèpre et illustrent l'application potentielle de la séro- prévalence comme un indicateur de l'amplitude de problème de la lèpre, au moins dans certaines zones.RESUMEN
Se hizo un estúdio para establecer si la seropreva- leneia de anticuerpos contra el glicolipidó fenólico-I de Mycobacteríum leprae (PGL-I) en los escolares, es un indicador útil de infección leprosa en determinadas áreas geográficas. El estúdio tiene la ventaja de poderse enfocar a una población fácilniente accesible, estable y estandarizada. Los anticuerpos anti-PGL-I se midieron usando la prueba simple de aglutinación de partículas de gelatina. Determinamos las tases de prevalencia en 2835 escolares de 5 diferentes áreas de 3 províncias de Sulawesi, Indonésia. Très áreas con una tasa de detección de casos de 3,4/10,000 se designaron como áreas de alta endemia. Las otras dos se designaron como áreas de baja endemia con una tasa de detección de casos de menos de 1/10,000. La seroprevalencia de anticuerpos en las 3 áreas de alta endemia varió dei 36% al 28% (95% Cl 21 %-31 %). En las dos áreas de baja endemia la tasa de prevalencia fue dei 7% (95% Cl 5%-10%). En un segundo estúdio efectuado 3 anos después en una de las áreas de alta endemia, la seroprevalencia fue la misma que la encontrada en el primer estúdio. Los resultados indican que las tasas de seroprevalencia entre los escolares pueden reflejar la incidência de lepra e ilustran la potencial aplicabilidad de la seroprevalencia como un indicador de la magnitud dei problema de la lepra en un área seleccionada.Mycobacterium leprae is the causative organism of leprosy, a disease which at present has a registered prevalence worldwide of about 830,000 people on antileprosy chemotherapy, although the estimated number of cases in the world reaches about 1.2 million (IS). The disease can cause permanent and extensive deformities of the skin and the peripheral nerves. Because of the severe and often irreversible handicaps and, as a consequence, social stigma and economic loss, the problem of leprosy is even greater than the number of cases would suggest.
The goal of the World Health Organization (WHO) is to eliminate leprosy as a public health problem by the year 2000, defined as a global prevalence of below 1 per 10,000. This program has resulted in intensified control activities, reducing the number of leprosy patients (16), but the end of the problem is not yet in sight. The effectiveness of the current strategy for routine leprosy control, based on case detection and treatment with multidrug therapy (MDT), has been questioned (6,10). With the present numbers of unevenly distributed leprosy cases and an incubation time of several years, many more leprosy patients can be expected to emerge in several leprosy-endemic countries beyond the year 2000 (2).
Information on the magnitude of the leprosy problem in any one area is important for the health services for their planning, monitoring and evaluation of leprosy control activities. The epidemiological data generated routinely by the health services will be greatly influenced by the policy and the activities of the services. These data do not necessarily reflect the true situation. Until now, the most reliable assessment of the leprosy problem in an area has been by random sample prevalence surveys. But such surveys are costly and cannot be carried out routinely.
Previous studies have suggested that the presence of IgM antibodies to phenolic gly- colipid-I (PGL-I) of M. leprae may be indicative of (past or present) infection (). In addition, it has been suggested that a high prevalence of anti-PGL-I IgM antibodies among children may be indicative of active transmission of M. leprae in their surroundings (1,4).
The aim of this study is to evaluate the validity of extrapolation of the seroprevalence in school children 10 to 12 years old as a proxy indicator for the level of leprosy endemicity. School surveys were chosen as the study design because they are relatively easy to perform and have the advantage of targeting an easily accessible, stable and standardized population.
MATERIALS AND METHODS
Study areas. With the objective of comparing seroprevalence rates with leprosy endemicity, we selected three provinces in Indonesia, each with different leprosy prevalences. They were South, North and South-East Sulawesi, with registered prevalences of 7.4, 3.5 and 2.1 per 10,000, and case detection rates of 3.9, 1.8 and 0.99/10,000, respectively.
Next, districts were selected. They had to be part of a well-established leprosy control program, with at least 5 years of 90%-100% implementation of MDT, to provide as reliable as possible estimates of the leprosy incidence. On the instigation of the leprosy control program, the following districts with different levels of endemicity were selected: in North Sulawesi, Minahasa district, with a case detection rate of 2.7/10,000; in South Sulawesi, Maros and Gowa districts with case detection rates of 2.1/10,000 and 5.1/10,000 respectively; in South-East Sulawesi, Muna district, with a prevalence of 1.4 per 10,000. The case-detection rates cited are the averaged rates for the 3 years prior to the year in which the school survey was conducted to reduce the effect of year-to-year variations.
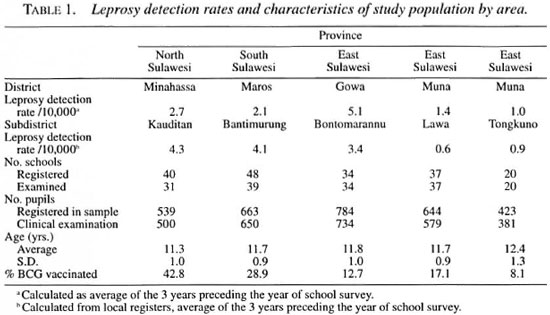
In each district, a subdistrict was chosen as the reference area for the school surveys. The selection of the subdistrict was not random but, again, based on the recommendations of the control program for the most reliable leprosy estimates. The subdistricts were: Kauditan in Minahasa, Bantimurung in Maros, Bontomarannu in Gowa, and Lawa and Tongkuno in Muna.
All patients from the local registers were entered in a computerized database using the Epi-Info software. Total detection rates were calculated per subdistrict. Patients who were actively detected in mass and focal surveys were excluded to minimize the influence of different control activities on the number of patients registered. The leprosy case-detection rates of the preceding 3 years were averaged and the result was used as a proxy indicator of the leprosy endemicity for that area.
School surveys. The school surveys were carried out between 1994 and 1998. In each subdistrict, all primary schools with a grade 5 were listed. In the subdistricts Kauditan and Bantimurung, the schools were selected using single-stage cluster sampling with a probability proportionate to size allocation scheme (5). Sample size calculations were based on an expected prevalence of 30% with an error of 5% and a design effect of 1.5. Expectations were based on previous community-based studies. In the other districts, all listed schools were enrolled in the study, since the required sample size was close to the total number of students registered.
All grade 5 pupils at the listed schools were included in the study. Each child present at the school during the survey was examined clinically by experienced local leprosy workers for signs of leprosy. Information on BCG vaccination status was obtained by visual inspection for the presence of a BCG scar. Household leprosy contact status was asked for and verified with the local records. Finger-prick blood was collected and serum separated from it on the same day. The serum was frozen at -20ºC until testing. In Bantimurung subdis- trict, a second school sero-survey was carried out 3 years after the first.
Serology. Antibodies to PGL-I of M. leprae were detected in a simple gelatin particle agglutination test (MLPA Serodia- leprae kit; Fujirebio, Japan) (8). This test will be referred to as MLPA. Sera with a titer of > 1:32 were considered positive.
Data analysis. All data were entered on a personal computer and analyzed using Epi-Info version 6.04. Analyses of variance were applied as indicated in the text. All probabilities presented are two-tailed, unless otherwise stated. The heterogeneity of seroprevalence among the different school clusters was examined with a goodness-of- fit test (Σ[(0 - E)2/E]).
RESULTS
Leprosy indicators. Examination of the local records revealed that the incidence rates at the level of the subdistricts showed a different pattern in comparison with the districts and the provinces (Table 1). Based on the local data, we divided the subdistricts involved into regions of different endemicity: three areas, Kauditan, Maros and Gowa, with a case detection rate of at least 3.0/10,000, will be referred to as the high- endemic regions. Two areas, Lawa and Tongkuno, with a case-detection rate of less than 1/10,000, were considered the low-endemic regions.
School surveys. The characteristics of the schools included in the surveys are shown in Table 1. For the first surveys, from the total of 3203 children registered at the different schools 2844 (89%) were included in the sample and examined. Sera were collected and tested from 2835 children (89% of the number registered).
The male-to-female ratio was not significantly different among the subdistricts (chi-squared, p = 0.21). The mean age of the school children was also similar in the three subdistricts.
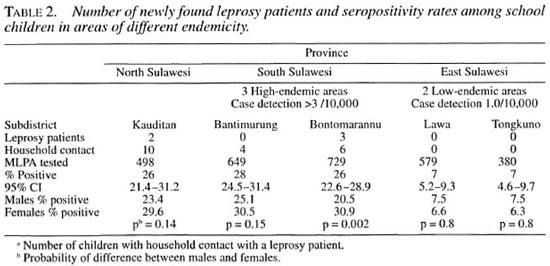
In the high-endemic areas, five children were newly diagnosed with leprosy during the survey, and 20 children reported having a member of the household with clinical leprosy (Table 2).
The BCG vaccination coverage was highest in the high-endemic area; it ranged from 42.8% there to 8.1% in one of the low-endemic areas.
Serology. Table 2 shows the results of the serological examinations. The seropositivity rates in the three high-endemic areas were similar, ranging from 26% to 28%. They were notably higher than the 7% found in the low-endemic areas. The correlation coefficient between incidence and seroprevalence was 0.98.
A significant difference in seroprevalence was found between male and female subjects in the high-endemic areas, but not in the low-endemic areas (Table 2).
To look for a possible age effect, the school children were divided into two groups: under 12 years of age and 12 years or older. No difference was found in the seropositivity rates between these two age groups (results not shown).
No relation was found between seropositivity and BCG status.
To investigate the heterogeneity of scro- prevalence in the different school clusters, the expected prevalence rate in each cluster was calculated and compared with the observed prevalence rate. Seropositivity was clearly uniformly distributed in all school clusters in four of the five areas (p >0.3). However, in Bantimurung subdistrict, a high-endemic area, we observed a higher seropositivity rate than expected in two of the 39 schools (p <0.005 and p = 0.04, respectively), which brought the assumption of heterogeneity of the clusters to a probability of p = 0.07.
In the second survey in Bantimurung, 3 years after the initial survey, 905 children from grade 5 were examined. The scro- prevalence of 28% was exactly the same as that found in the first survey. The leprosy case detection rate was also similar 3 years later.
DISCUSSION
A prerequisite for any disease control program is to be able to measure, or at least to estimate, the prevalence of the disease. Such information is essential not only for the allocation of resources, but also for evaluating the effect of the control measures. At present, leprosy control programs face difficulties in estimating the burden of disease. Because leprosy prevalence is generally low, accurate assessment of the prevalence of the disease can only be obtained through laborious and expensive mass surveys. Official data from registers are often not reliable, since they are greatly influenced by the policies and the quality of the local or national leprosy control program. We have looked at an alternative approach to assess the magnitude of the leprosy problem, extrapolation of the seropositivity rates for the species-specific antigen PGL-I in school children. The results suggest that this seropositivity is related to the leprosy incidence in that population. This approach may thus provide a rapid, reliable and relatively inexpensive method to estimate the leprosy burden in a selected area.
We have determined the seroprevalence rates in school children in five selected areas in Sulawesi, Indonesia. Three had similar high leprosy case-detection rates of 3.4 to 4.3/10,000 and seroprevalence rates of 26% to 28%. The other two regions had much lower incidence rates of less than 1.0/10,000 and also lower seropositivity rates (7.1%). It should be noted that these case-detection rates are based on passive case finding only.
In our opinion, it is valid to compare these rates from the different areas, since the control programs active in these areas are under the same management policies. However, our data also illustrate that it is an almost impossible task to obtain true incidence rates in a leprosy-endemic area. At best, the case detection rates can be used, which we have also done in our studies, but these rates are highly dependent on the commitment of the local leprosy workers. In addition, the figures reported represent aggregated data which may completely obscure pockets of high leprosy endemicity such as those we have encountered in North Sulawesi. Although this province was classified as a low-endemic area and very few cases had been reported in the past years from the selected subdistrict, the examination of the local registers revealed the subdistrict to be highly endemic. The low numbers of patients reported may have been caused by a change of the local leprosy health worker followed by a temporary absence of one. The load of undetected cases and, therefore, the difficulty in obtaining a true picture of the leprosy burden is further illustrated by the high number of leprosy patients who were detected actively in the occasional mass surveys. Nevertheless, we feel confident that the designation of the studied areas into high- and low-endemic areas is justified. We have presented the average case detection rates over the last 3 years, yet inspection of the data of previous years gave a similar ranking of the areas.
A relation between the leprosy burden and seroprevalence in different parts of the world has been documented by several other studies (4,11,12). In addition, Baumgart, et al. (1) found that, upon introduction of MDT in a closed community in Papua New Guinea, the decrease in leprosy prevalence was accompanied by a reduction in the seropositivity rate among children. Unfortunately, a valid comparison of either seropositivity or leprosy prevalence rates between these studies is not possible, since different methods were applied to obtain the data. We have already discussed the difficulties in comparing the official data on leprosy prevalence. Comparison of serological results is only possible when the same standardized test has been used. We have therefore used a commercially available, standardized assay in kit format (8) which guarantees comparable results.
Since seropositivity rates have previously been shown to be age-dependent (7,14), we chose school children as the study population. This approach offers the combined advantages of having an age-standardized population and a relatively easy access to the study population when conducting the surveys. Only in the high-endemic areas did we find a higher seropositivity rate among females, as has been reported for older age groups (7,14). This may reflect a more avid response in females upon repeated exposure. The rate of positive BCG vaccination status was highest in the high-endemic areas. Clearly, risk factors other than BCG status are predominant in determining the development of leprosy in these areas.
One of the features of the epidemiology of leprosy can be its clustered distribution 03). However, in four of our areas, the seropositivity rates were uniformly distributed in all clusters, suggesting that seropositivity reflected an endemic exposure to M. leprae on a population level. In only one area did one particular school cluster show an unexpectedly high seroprevalence rate. From a total of 161 school clusters the number having only one homogeneous cluster is so low that it does not influence the applicability of estimating seroprevalence by using a single-stage cluster design with school as sampling units. Heterogeneity of seropositivity in populations at the village level in Sulawesi has been reported before (14). A second survey conducted more than 3 years later showed exactly the same seroprevalence rates, revealing the consistency of seropositivity which was paralleled by a consistency in case-detection rates.
Although the leprosy detection rate and seroprevalence showed a strong correlation, the number of areas studied is too small for firm numerical conclusions. We also lacked an area with an intermediate endemicity. It is, therefore, not possible to predict whether the nature of the relationship will be linear or logarithmic in nature.
We propose to validate the approach to assessment of the leprosy problem through extrapolation of seropositivity rates in school children in other areas, both in Indonesia and elsewhere in endemic countries. In this study we used a standardized particle agglutination assay (MLPA) for the detection of antibodies to PGL-I, a technique which still requires a laboratory facility. We have now developed a simple dipstick assay to detect antibodies to PGL-I of M. leprae which can be performed in the field and does not depend on a cold chain (3). Using this method, together with a simplified sampling frame, may make rapid epidemiological mapping of leprosy a feasible prospect in the near future.
Acknowledgment. We express our gratitude to the heads of the Offices of the Ministry of Health, Republic of Indonesia, in Sulawesi and to the leprosy officers in the Ministry of Health and in the provinces of Sulawesi for their cooperation. We are obliged to Dr. Peter Lever and Dr. Frankie Lopreng for their valuable advice. We thank Mr. Romi and Mr. Marrwani for excellent technical assistance. We are especially indebted to all the school children and their parents who cooperated voluntarily in this study. The financial support by the Netherlands Leprosy Relief Association (NSL) is greatly appreciated.
REFERENCES
1. Baumgart, K. W., Britton, W. J., Mullins, R. J., Basten, A. and Barnetson, R. St.C. Subclinical infection with Mycobacterium leprae -a problem for leprosy control strategies. Trans. R. Soc. Trop. Med. Hyg. 87(1993)412-415.
2. Becx-Bleumink, M. Success of the WHO multidrug therapy? Trop. Geogr. Med. 6 (1994) 61-64.
3. Buhrer, S. S., Smits, H. L., Gussenhoven, G. C., van Ingen, C. W. and Ki.atser, P. R. A simple dipstick assay for the detection of antibodies to phenolic glycolipid-I of Mycobacterium leprae . Am. J. Trop. Med. Hyg. 58 (1996) 133-136.
4. Cho, S.-N., Kim, S. H., Cellona, R. V., Chan, G. P., Fajardo, T. T., walsh, G. P. and Kim, J. D. Prevalence of IgM antibodies to phenolic glyco- lipid I among household contacts and controls in Korea and The Philippines. Lepr. Rev. 63 (1992) 12-20.
5. Division for the Control op Diarrhoeal and Acute Respiratory Disease. Diarrhoea morbidity, mortality and treatment practices: household survey manual. Geneva: World Health Organization.
6. Fine, P. E. M. Reflections on the elimination of leprosy. Int. J. Lepr. 60 (1992) 71-80.
7. Fine, P. E. M., Ponnighaus, J. M., Burgess, P., Clarkson, J. A. and Draper, C. C. M. Seroepi- demiological studies of leprosy in northern Malawi based on an enzyme-linked immunosorbent assay using synthetic glycoconjugate antigen. Int. J. Lepr. 56 ( 1988) 243-254.
8. Izumi, S., Fujiwara, T., Ikeda, M., Nishimura, Y., Sugiyama, K. and Kawatsu, K. Novel gelatin particle agglutination tests for serodiagnosis of leprosy in the field. J. Clin. Microbiol. 28 (1990) 525-529.
9. Klatser, P. R. Serology of leprosy. Trop. Geogr. Med. 46 (1994) 59-62.
10. Leprosy beyond the year 2000. (Editorial) Lancet 350 (1997) 90-93.
11. Shah, D. H., Vartak, R. R., Naik, S. S., Dan- dekar, S. R. and Ganapati, T. Epidemiological studies in children of a low-endemic region, a high-endemic region, and dwellers of a leprosy colony: evaluation of anti-ND-BSA antibodies and lepromin responses. Int. J. Lepr. 62 (1994) 539-546.
12. Ulrich, M., Smith, P. G., Sampson, C., Zunioa, M., Centeno, M., Garcia, V., Manrique, X., Salgado, A. and Convit, J. IgM antibodies to native glycolipid-I in contacts of leprosy patients in Venezuela: epidemiological observations and a prospective study of the risk of leprosy. Int. J. Lepr. 59 (1991) 405-415.
13. van Beers, S. M., de Wit, M. Y. L. and Klatser, P. R. The epidemiology of Mycobacterium leprae; recent insight. FEMS Microbiol. Lett. 136 (1996) 221-230.
14. van Beers, S. M., Izumi, S., Madjid, B., Maeda, Y., Day, R. and Klatser, P. R. An epidemiological study of leprosy infection by serology and polymerase chain reactions. Int. J. Lepr. 62 (1994) 1-9.
15. World Health Organization. Progress towards the elimination of leprosy as a public health problem. Wkly, Epidemiol. Rec. 70 (1995) 177-182.
16. World Health Organization. Trends in leprosy detection. Wkly. Epidemiol. Rec. 73 (1998) 169-176.
1. Ph.D., Department of Biomedical Research, Royal Tropical Institute, Meibergdreef 39, 1105 ZA Amsterdam, The Netherlands.
2. Ph.D., Department of Biomedical Research, Royal Tropical Institute, Meibergdreef 39, 1105 ZA Amsterdam, The Netherlands.
3. M.D., Ph.D., Department of Microbiology, Hasanuddin University, Ujung Pandang, South Sulawesi, Indonesia.
Reprint requests to Dr. Klatser at the above address or e-mail: P.Klatser@kit.nl
Received for publication on 30 March 1999.
Accepted for publication on 12 May 1999.