- Volume 66 , Number 3
- Page: 348–55
Quantitative assessment of facial sensation in leprosy
ABSTRACT
The trigeminal and great auricular nerves which supply sensation to the face are affected in leprosy. No objective sensory testing methods have been devised for testing sensation in the face. Testing for corneal sensation to ascertain trigeminal nerve or visualization and palpation of the great auricular nerve alone may not be enough to establish the involvement of these nerves.In a sample of leprosy patients, face sensation threshold measurements were done using a set of three Semmes-Weinstein (SW) monofilaments that gave a force of 0.05-0.07, 0.2 and 2 g. Sensation was tested by three examiners and intra- and inter-observer testing was used as a means to validate the findings.
Within the limitations of this study, the results indicate that use of SW monofilaments is a fairly reliable and repeatable method for sensory testing in the face. During follow up, a single filament with a force of 0.5-0.7 g (2.83 marking number in SW filament or any other filament with a corresponding gram force) could be used to assess sensation. A simple procedure of quantifying sensation in these nerves is suggested. A method to incorporate trigeminal or great auricular nerve sensory testing into the existing sensory assessment charts is also discussed.
RÉSUMÉ
Les nerfs sensitifs trijumeaux et grands auriculaires sont souvent affectés durant l'évolution de la lèpre. Un test objectif de sensibilité n'a pas encore été développé pour évaluer la fonction nerveuse sensitive du visage. L'évaluation du nerf trijumeau en testant la sensibilité coméenne ou las visualisation et la palpation du nerf grand auriculair ne sont peut-être pas suffisant pour démontrer l'atteinte de ces nerfs.A partir d'un échantillon de patients atteints de lèpre, des mesures de sensibilité minimale du visage furent effectuées en utilisant un ensemble de trois monofilaments de Semmes-Weinstein (SW) qui produisent une force de 0,05-0.07; 0.2 et 2 g. La sensibilité fut testée par trois examinateurs et des tests intra- et in ter-observateurs furent utilisés pour valider les données observées.
Malgré les limitations de cette étude, les résultats suggèrent que l'utilisation de monofilaments de SW est une méthode tout à liât fiable, ayant une bonne répétabilité, [?| pour tester la sensibilité du visage. Durant le suivi, un seul filament avec une force de 0,5-0,7 g (type 2,83 pour les filaments de SW ou n'importe quel autre filament ayant une force en grammes correspondante) pourrait être utilisé pour évaluer la sensibilité. Une procédure simple pour quantifier la sensibilité de ces nerfs est proposée. Une méthode est aussi discuttée pour intégrer les tests de sensibilité des nerfs trijumeau et grand auriculaire dand les schémas déjà existants d'évaluation de la sensibilité.
RESUMEN
Los nervios trigémino y gran auricular que proporcionan sensibilidad a la cara están afectados en la lepra. Sin embargo, hasta ahora no se han desarrollado métodos objetivos para medir la sensibilidad de la cara. Las pruebas de sensibilidad de la córnea para establecer la función del trigémino y la palpación del nervio gran auricular pueden resultar ¡nsulicientes para establecer la afección de estos nervios.En este estudio se midió el umbral de sensibilidad de la cara en un grupo de pacientes con lepra usando una serie de 3 monolilamentos de Semmes-Weinstein (SW) calibrados para ejercer una presión de 0.05-0.07. 0.2 y 2 g. La sensibilidad se probó por 3 examinadores y los resultados y variaciones entre ellos se usaron para validar las mediciones.
Dentro de las limitaciones del estudio los resultados indicaron que el uso de los monolilamentos de SW constituye un método confiable para medir la sensibilidad de la cara. Durante el seguimiento, la sensibilidad podría establecerse usando un solo filamento con una fuer/a de 0.5 a 0.7 g (filamentos SW o cualquier otro del número 2.83). Se sugiere un método simple para medir la sensibilidad de estos nervios y se discute la posibilidad de incorporar la prueba sensorial de los nervios trigémino y gran auricular dentro de las pruebas de sensibilidad existentes.
Several methods exist for sensory testing of the hands and feet of leprosy patients (2, 3, 10, 13, 17). Normal sensation of the human face has been documented (4, 18, 20) and methods to systematically test sensation in the face in the post-traumatic population has been documented (12), but no such testing method has been formalized in the leprosy literature despite the fact that the trigeminal nerve and the great auricular nerve, which supply sensation to the face, are affected in this disease.
At present, great auricular nerve involvement is ascertained by visually checking for nerve enlargement and by testing for tenderness by palpation (8, 11). Trigeminal nerve involvement cannot be checked using this method since it is situated in a shallow bony depression at the apex of the petrous bone. However, one of its branches, the supraorbital nerve, when thickened, is visible and palpable (8). Assessment of corneal sensibility is the only other available method to detect involvement of the trigeminal nerve.
Knowing about facial hypesthesia becomes important, firstly, when a person may possibly have an involved great auricular nerve and/or trigeminal nerve in the absence of other signs. In this case, the presence of a facial sensory deficit will probably clinch the diagnosis of leprosy. Secondly, if the person had only one skin patch and, possibly, an involved trigeminal and/or great auricular nerve, the finding of facial hypesthesia would make the difference between single-dose ROM (rifampin + ofloxacin + minocycline) and 6-month PB MDT (paucibacillary multidrug therapy). Nevertheless, in many other patients with other definite signs of leprosy or a positive skin smear, the presence or absence of facial hypesthesia would not make any difference in the clinical diagnosis, but it can be useful in the clinical management by monitoring the status of these nerves. Thirdly, it might be useful for the evaluation of treatment of leprosy.
This study describes a quantitative method of assessing sensation in the face using Semmes-Weinstein monofilaments. Intraand inter-observer reliabilities were assessed to determine whether this method can be used to evaluate changes in sensation of the face.
SUBJECTS AND METHODS
Leprosy patients attending the outpatient department of the Schieffelin Leprosy Research and Training Centre, Karigiri, India, during July 1994 were screened. A nonrandom sample of 13 patients was selected based on the following criteria decided by a consensus between two experienced leprologists: a) representation from all clinical classifications, b) an equal distribution by sex as far as possible, and c) an equal distribution of patients with or without facial patches. There were five males and eight females. Their clinical classifications were: 3 tuberculoid, 3 borderline tuberculoid, 4 borderline borderline, 2 borderline lepromatous and 1 lepromatous leprosy. Six patients with visible facial patches and seven without visible patches on the face were selected for inter-observer testing, which gave a total of 26 pairs of nerves for testing. Among these 13 subjects, the first 5 patients with visible facial patches and 5 without visible patches on the face were selected for intra-observer testing.
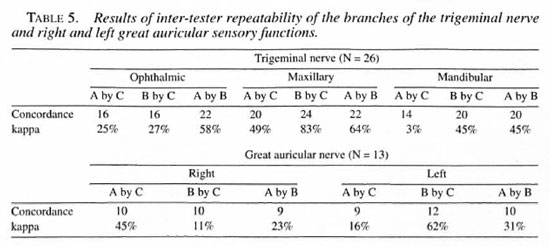
Testing sites. Twenty-three testing points were selected on the face (Fig. 1): 6 points (1-6) from the area supplied by the ophthalmic branch of the trigeminal nerve, 6 points (7-12) from the maxillary branch, and 7 points (13-19) from the mandibular branch. Four points (20-23) were selected from the area supplied by the great auricular nerve.
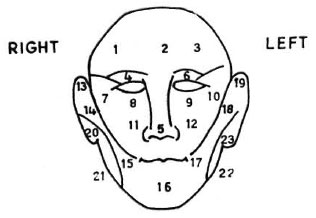
Fig. 1. Twenty-three facial points for testing facial sensation.
We wanted to select 10 standard testing sites from the above 23 for the purpose of incorporating them into the existing sensory assessment chart. Sensory evaluation was done and the subjects with facial anesthesia were identified during testing and re-testing by three assessors (A, B, C). In selecting the 10 standard sites, no reduction was made from the original testing points for the great auricular nerves (four sites). The remaining six sites were allotted to the trigeminal nerve by choosing one point for each branch (ophthalmic, maxillary and mandibular). The selection of sites for each branch was based on the location in which maximum sensory loss was elicited among the 3-4 sites that were examined for that branch.
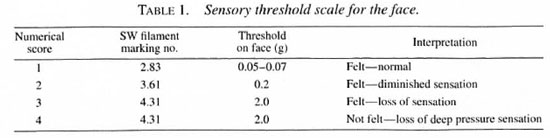
Testing instrument. The lightest three filaments in a Semmes-Weinstein (SW) filaments "mini" kit (1) were used for this study. The filaments have a "bending force" of 0.05-0.07, 0.2 and 2 grams, respectively. However, the 2-g force which indicates "diminished protective sensation" for the hand was considered "loss of sensation" for the face, and the 0.2-g filament which indicates "diminished light touch" for the hand was considered "diminished sensation" for the face. The sensory thresholds used are given in Table 1.
Testing method. The thinnest filament (No. 1) was used first on the 23 test sites with the patient's eyes closed. It was bent gently on the surface of the skin, held for 1 to 2 seconds, and then lifted slowly. This was repeated three times for each site. The patient was asked to point with a finger to the stimulated site. If the patient pointed at least two times within 2 cm of the stimulated point, the sensation of the site was considered normal. If there was no adequate response, the next heavier filaments (Nos. 2 and 3) were applied successively.
Wind and noise may distract the patient and, therefore, testing was done in a quiet room with the ceiling fan switched off. Testing was performed by three independent assessors with the professional background of leprosy paramedical work, optometry and occupational therapy.
All tests were carried out within 2 days. A resting time of 15 min was allowed between assessments to avoid possible fatigue which might result in a decrease in attention span which could influence test results. For the intra-observer reliability study, the first test results were collected from the observers immediately after the assessments were done. Re-testing was done on the same patient only after several other patients had been examined. This was to avoid the possibility of recall bias.
Statistics. Intra- and inter-observer reliability was assessed in three stages; 1) an overall score of sensation of the face, 2) a score for each of the two nerves, and 3) a reliability score for the three branches of the trigeminal nerves for each site on the face.
The kappa coefficient was used to determine the extent of chance-corrected intraand inter-observer agreement on a scale of 0-100%. A value below 40% indicates poor agreement; 40% to 75%, fair to good; values >75%, excellent agreement (7). An example of the agreement matrix is given in Table 2.

RESULTS
Facial anesthesia was present in 8 of the 13 patients studied. For these 8 patients 45 sensory assessments were available, giving a total of 1035 sites for analysis. Loss of sensation was present in the following 10 points from the 45 records: 1) right upper lid, 11 records; 2) left upper lid, 13 records; 3) right lower lid, 6 records; 4) left lower lid, 8 records; 5) right pretragus area, 5 records; 6) left pretragus area, 9 records; 7) right lower tip of ear, 20 records; 8) right angle of mandible, 6 records; 9) left angle of mandible. 5 records; and 10) left lower tip of ear, 3 records.
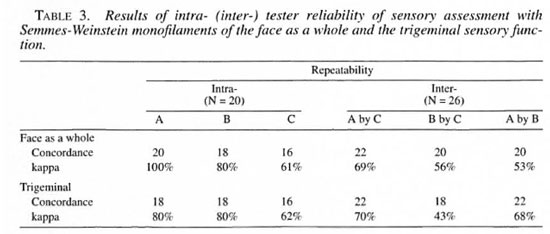
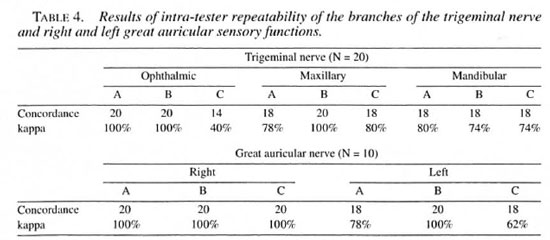
The intra-observer variation in the face as a whole, the trigeminal nerve and its branches, and the right and left great auricular nerves ranged from 40% to 100% (Tables 3 and 4). The inter-observer variation in the face as a whole and trigeminal nerve ranged from 43% to 7()7r> (Table 3). The same variation for the three branches of the trigeminal and the right and left great auricular nerves ranged from 3% to 83% (Table 5).
DISCUSSION
Quantitative sensory testing of the face has not been given much attention in the early detection of leprosy and evaluation of treatment for leprosy. The main reason is because the face, unlike the hands and feet, does not directly interface with the environment and is, thus, unlikely to be traumatized if anesthetic. Since there are reports of ocular complications occurring in association with facial patches (9), the value of determining sensation in the face becomes important, and may also be a necessary procedure for the evaluation of the great auricular and trigeminal nerve involvement.
Consistency of testing. The good consistency in the intra- and inter-observer agreement in the major nerve trunk and the intra-observer agreement while investigating its smaller branches indicate that this method can be advocated for the evaluation of sensory changes in the face. Inter-agreement did not reach acceptable values in the smaller branches of the nerve trunks. It seems strange that a concordance of 14 out of 26 for the trigeminal nerve would produce a kappa value of 3%. This problem is well known and occurs with certain distributions of the marginal totals of agreement matrixes (6). In this study symmetrical imbalanced marginals, as shown in Table 2, resulted in a very low kappa value, despite the fact that the direct agreement had a value of 53.8%, and this could explain the difference in kappa value. More training and practice may be required to reduce such inter-observer variations while testing smaller branches of nerves in the face. Since we lacked a "gold standard" and since this test was being done for the first time, further studies are needed to overcome this problem.
Trigeminal nerve involvement. The trigeminal nerve consists of a motor and a sensory division. In leprosy only the sensory division is affected, resulting in impaired sensation of the face. Palpating the supraorbital nerve and checking for corneal sensation assesses only the ophthalmic branch of the trigeminal nerve. The other two branches of the nerve usually go unexamined. Facial sensory testing as suggested will give quantitative sensory information for all three branches of the trigeminal nerve (see Annex).
The sensation of the mucosal surface of the eye is supplied by the trigeminal nerve (16), and evaluation of changes in sensation of the face, particularly around the eyes, could indicate corneal insensitivity. This hypothesis can be tested in a further study.
Great auricular nerve involvement. Dharmendra and Chatterjee stated that 18.9% of leprosy cases have enlarged great auricular nerves (5). The nerve is normally examined by asking the patient to turn the head to the opposite side. A thickened nerve can then be seen or palpated across the sternocleidomastoid muscle. A study on 175 healthy young soldiers showed that in 67% the great auricular nerve was palpable and in 39% it was both visible and palpable (15). This could mean that enlargement of the nerve does not necessarily indicate great auricular nerve involvement in leprosy patients. In the absence of other cardinal signs of leprosy, hypesthesia in the area distributed by the nerve would be indicative of the disease.
Selection of filament to test the face.
A few testing instruments are in common use to assess and grade sensory function in leprosy patients. The World Health Organization (WHO) suggested sensory testing with a ballpoint pen; other instruments used include the Semmes-Weinstein monofilaments, a tuning fork or a biothesiometer vibrating at 120 Hz, and a paperclip or diskcriminator for 2-point discrimination. The filaments became increasingly accepted in leprosy and have been field tested by many workers in India (10), Nepal (3), Thailand (13), the U.K. (17), the U.S.A. (2), and in Ethiopia (14). These studies have shown that the filaments have good repeatability in clinical testing.
The SW filaments are available in a "mini" kit which contains five filaments and a "long" kit with 20 filaments (1). The "mini" kit contains the three filaments that have been used in this study. These kits are now readily available in leprosy-endemic countries (2, 10).
We selected the 2.83 SW filament (approximately 0.05-0.07-g bending force) to test the lowest threshold of pressure sensation because tactile sensitivity of the face among normal persons in earlier studies had shown the mean threshold of males to be 0.02 g; females, 0.018 g (20). Using a filament that gives a force of 0.05-0.07 g will be higher than the threshold for the face and will avoid false negative responses for the following reasons: a) the lowest sensory threshold in normal individuals quoted in the above article is in the laboratory situation. The study of Costas, el al. has shown that the 2.44 SW filament, giving a force of 0.0346 g/mm2, was not felt at the upper eye lids and the pretragus areas by 1.6% of the normal population (4), which indicates that in a clinical situation the next higher threshold may be required to increase the test sensitivity, b) Posnick, et al. showed that the facial threshold approximated similar sensory thresholds for the index fingertip (18). SW filament 2.83 is the first choice in testing normal sensation of the hands (1,2).
Standardization of testing sites. The original 23 testing sites (Fig. 1) could be reduced to 10 for routine clinical examination, as shown in Figure 2. These sites represent all nerve branches that supply sensation to the face. These 10 points on the face can then be standardized along with the existing pattern of examining sensation in 10 sites on the hands and feet (Fig. 2). A method to calculate the score for each nerve is described in the Annex.
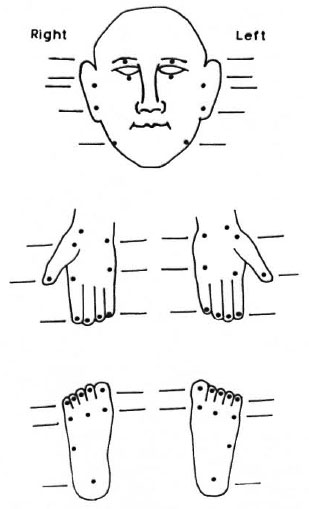
Fig. 2. Suggested 10 sites for routine clinical testing of sensation on the face to match existing 10 sites each on the hands and feet.
Modifications to commonly used sensory assessment charts are suggested. One is the sensory impairment record as suggested by Lienhardt, et al. (14) which could be modified as shown in Figure 2. This can be used as the basic record for detailed assessment. We suggest the use of the same three filaments used in this study for the face. Second is the field-based "Disability Continuation Record" as suggested by Watson (19) which has been adopted in several centers (Figure 3 illustrates a modification of this). In a field situation there is probably no need for the routine examination of facial sensation. Therefore, we recommend that in this disability record the face be assessed less frequently in comparison to the hands and feet, using a single filament (0.05-0.07-g force) for the face.
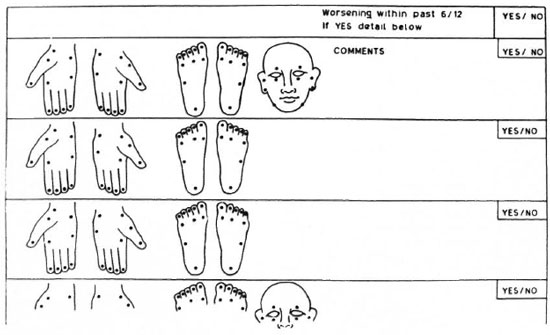
Fig. 3. Modification of currently used, field-based "Disability Continuation Record" suggested by Watson (19).
Acknowledgment. We thank Dr. P. S. S. Sundcr Rao. Director, Schieffelin Leprosy Research and Training Centre (SLR&TC), for his counsel; Ms. Nisha Kurian, Biostatistician, SLR&TC, for statistical assistance; and Dr. J. W. Brandsma, Physical Therapist, National Institue for Research & Postgraduate Education. Aniersfoort. The Netherlands, for comments and advice.
REFERENCES
1. BELL-KROTOSKI, J. Advances in sensibility evaluation. Hand Clin. 7 (1991) 527-546.
2. BELL-KROTOSKI, J., FESS, E., FIGAROLA. J. H. and HILTZ, D. Threshold detection and Semmes-Weinstein monofilaments. J. Hand Ther. 9 (1995) 115-162.
3. COSTAS, P. D., HEATLEY. G. and SECKEL, B. R. Normal sensation of the human face and neck. Plast. Reconstr. Surg. 93 (1994) 1141-1145.
4. DHARMENDRA and CHATTERJEE, S. N. Examination for detection of thickened nerves. In: Leprosy, Volume I. Dharmendra. ed. Bombay: Kothari Medical Publishing House, 1985. pp. 251-252.
5. FEINSTEIN, A. R. and CICCHETTI, D. V. High agreement and low kappa. I. The problem of two paradoxes. J. Clin. Epidemiol. 43 (1990) 543-549.
6. FLEISS, J. L. The measurement of interrater agreement. In: Statistical Methods for Rates and Proportions. 2nd ed. New York: John Wiley & Sons, 1981, pp. 211-263.
7. HOGEWEG, M.. KIRAN, K. and SUNEETHA, S. The significance of facial patches and type 1 reaction for the development of facial nerve damage in leprosy; a retrospective study among 1226 paucibacillary leprosy patients. Lepr. Rev. 62 (1991) 143-149.
8. JERSKEY, R. S. Semmes-Weinstein monofilament testing to determine normal sensory thresholds in the foot and hand: a comparative study in India. (Abstract) Int. J. Lepr. 61 Suppl. (1993) 159A.
9. JOPLING, W. H. and McDOUGALL, A. C. CLINICAL aspects. In: Handbook of Leprosy. 5th edn. New Delhi: CBS Publishers. 1996, p. 25.
10. KESARWANI, A., ANTONYSHYN. O. P.. MACKINNON, S. E.. GRUSS, J. S.. NOVAK, C. and KELLY, L. Facial sensibility testing in the normal and post-traumatic population. Ann. Plast. Surg. 22 (1989) 416-420.
11. KUIPERS, M. and SCHREUDERS, T. The predictive value of sensation testing in the development of neuropathic ulceration on the hands of leprosy patients. Lepr. Rev. 65 (1994) 253-261.
12. LlENHARDT, C, CURRIE, H. and WHEELER, J. G. Inter-observer variability in the assessment of nerve function in leprosy. Int. J. Lepr. 63 (1995) 62-76.
13. LYNCH. P. and JOHNSON, J. H. The great auricular nerve in presumably non-leprous British soldiers. Trans. R. Soc. Trop. Hyg. 74 (1980) 136.
14. MUMENTHALER, M. General neurological principles. In: Neurology. 5th edn. Chicago: Year Book Medical Publishers, Inc.. 1977, p. 14.
15. OWEN, B. M. and STRATFORD, C. J. Assessment of the methods available for testing sensation in leprosy patients in a rural setting. Lepr. Rev. 66 (1995) 55-62.
16. PFALTZXGRAFF. R.E. and RAMU', G. Clinical leprosy. In: Leprosy. 2nd edn. Hastings, R. C., ed. Edinburgh: Churchill Livingstone. 1994, p. 243.
17. POSNICK, J. C. ZlMBLER, A. G. and GROSSMAN, J. A. I. Normal cutaneous sensibility of the face. Plast. Reconstr. Surg. 86 (1990) 429-433.
18. VAN BRAKEL, W. H., SHUTE, J.. DlYON, J. A. and ARZET, H. Evaluation of sensibility in leprosy- comparison of various clinical methods. Lepr. Rev. 65 (1994) 106-121.
19. WATSON, J. M. Preventing disability in leprosy patients. London: The Leprosy Mission International. 1988. pp. 33-34.
20. WEINSTEIN, S. Intensive and extensive aspects of tactile sensitivity as a function of body part, sex and laterality. In: The Skin Senses. Kenshalo, D. R., ed. Springfield, IL: Charles C. Thomas Publishers. 1968. pp. 193-218.
1. Ph.D.. Department of Occupational Therapy and Health Education.
2. M.B.B.S., M.S. (Ophth.). Branch of Ophthalmology.
3. M.B.B.S.. D.C.P., Department of Immunology and Clinical Pathology.
4. Branch of Ophthalmology. Schieffelin Leprosy Research and Training Centre. Karigiri 632 106. Tamil Nadu. India.
Received for publication on 5 February 1998.
Accepted for publication in revised form on 6 May 1998.
ANNEX
Method to Score Sensory Nerves Supplying the Face
Three testing points have been identified on each side of the face for the trigeminal nerve. If the patient feels a 0.05-0.07-g filament on each point, a score of 0 is given to that site, a score of 1 for not feeling that filament, etc. Thus, total loss of sensation at a point will be scored as 3. Since there are three testing points the maximum sensory loss per trigeminal nerve is scored as 3+3+3, a total of 9. Similarly, for the two sites for the great auricular nerve scoring would be 3+3, a total of 6. Normal sensation would be scored as 0 x 3 or 0 and 0 x 2 or 0. The table below shows the maximum score for total loss of sensation of the following nerves:
