- Volume 66 , Number 4
- Page: 485–7
Cellular immune responses of healthy and Mycobacterium leprae-lnoculated armadillos (Dasypus novemcinctus)
To the Editor:
The nine-banded armadillo (Dasypus novemcinctus) is one of the best experimental models for the study of leprosy, mainly because upon inoculation with live Mycobacterium leprae a disease similar to human lepromatous leprosy (LL) develops (6). Not all M. leprae-inoculated armadillos develop a systemic infection, up to 93% (5) and 60% (2) have been reported. Moreover, in most of the cases, after the experimental inoculation, an average of 1.5 to 3 years are needed before the presence of acid-fast bacilli (AFB) in lepromas, liver and spleen is observed (8 and Quesada-Pascual, et al, unpublished observations). On the other hand, it has previously been demonstrated that protection against the disease in humans depends upon the development of an appropriate cellular immune response to the bacillus (4). Therefore, in this study we analyzed the status of the cellular response in healthy and M. leprae-infected armadillos in order to correlate this immune response with the development of generalized infection in these animals. Lymphocytes from peripheral blood were stimulated in vitro with the polyclonal activator concanavalin A (ConA) and with soluble extracts from M. leprae (MLSE), M. habana (MHSE) and M. tuberculosis (MTSE) (prepared as described in 3).
Isolated peripheral blood mononuclear cells (PBMC) from 8 M. leprae -inoculated and 12 noninoculated armadillos were tested in a proliferation assay with MLSE, MHSE and MTSE as previously described (9). The results are shown in The Figure. None (0/12) of the armadillos in the noninoculated group showed any proliferation with MLSE, 4 (4/12) showed a mitogenic index higher than 3 with MHSE, and only 1 (1/11) was positive to MTSE. On the other hand, in the M. leprae -inoculated group, PBMC from 2 armadillos (2/8) showed a stimulation index higher than 3 when stimulated with MLSE; PBMC from 3 inoculated armadillos (3/6) were positive when tested with MHSE, but none (0/6) showed any proliferation with MTSE. There were statistically significant differences between the responses with MLSE (p < 0.05) and ConA (p < 0.05) for both the control noninfected group and the M. leprae -inoculated armadillos as analyzed by the Student's test. There were no significant differences between the responses with MHSE and MTSE (p > 0.05). Additionally, the infection was followed in all animals by macroscopic examination and by microscopic observation of Ziehl-Neelsen-stained smears for the presence of AFB as previously described (8). During the time of the study, only 3 armadillos showed abundant leprotics and were positive for AFB in liver and spleen imprints taken during autopsy. On the other hand, none of the other animals developed clear signs of infection, although some of them had small nodules that were all negative for AFB.
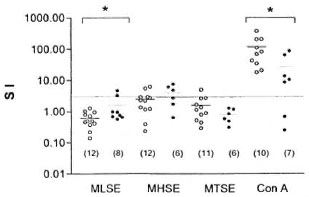
The Figure. Proliferation of armadillo peripheral blood lymphocytes induced hy soluble antigens from h M. leprae , M. habana and M. tuberculosis or concanavalin A (ConA). Lymphocytes from noninoculated armadillos (○) and M. leprae -inoculated armadillos (●) were placed isto 96-well, round-bottom plates at 4 x 105 cells/well. Soluble antigens from M. leprae (MLSE), M. habana (MHSE) and M. tuberculosis (MTSE) at 25 µg/ml or ConA at 12.5 µg/1111 were added to each well in a total volume of 200 µl. Control wells contained culture medium alone. Cultures were set up in triplicate and incubated for 3, 4, 5 and 6 days for ConA. MHSE, MTSE and MLSE, respectively, at 37°C in a fully humidified atmosphere containing 5% CO2. Proliferation was evaluated by 3H-thymidine in-corporation using 0.5 µCi/well during the last 12 hr of culture. The cells were then harvested onto glass-fiber filters with a semi-automatic sample harvester. 3H-thymidine incorporation was assessed by liquid scintillation spectroscopy. The results are expressed as astimulation index (SI) which is a ratio of the responses to MLSE, MHSE, MTSE and ConA to the control cultures. which contais neither antigen nor ConA. An índex equal to or higher than 3 was taken as a positive response. Each point represents a single a armadillo; numbers in parentheses are armadillos tested in each group; * = p < 0.05.
Our results showed that lymphocytes from most of the infected armadillos were unable to respond to soluble extracts of M. leprae, even though they had been exposed to the antigen while being inoculated. The absence of a lymphoproliferative response to MLSE in healthy animals may be explained by the fact that these armadillos were naive to these antigens. However, in our study lymphocytes from only 25% of the M. leprae -inoculated armadillos responded to M. leprae antigens. This lack of activity may correlate with the susceptibility of these animals to develop lepromatous-like leprosy, but our results do not correlate with the high percentage of armadillos developing systemic infection (2, 5). More studies are needed to clarify this point.
Although crossreactivity between mycobacteria has been shown (10, 13), there was no significant difference between the control group and the M. leprae -inoculated group in the responses with MHSE and MTSE. However, 4/12 of the noninoculated armadillos were positive to M. habana. This can be explained by the presence of crossreactive epitopes in mycobacteria which are present in the environment and have been in contact with the armadillos before their capture. Moreover, in our data no response is seen with MTSE for both M. leprae -inoculated animals and noninoculated animals. Similarly, in a study of eight-banded, noninoculated armadillos ( Dasypus sabanicola ) the responses of cell-mediated immunity to M. tuberculosis were very discrete; whereas delayed-type hypersensitivity and in vitro lymphoproliferation were usually negative in armadillos inoculated with M. leprae (12).
In our study, there was a significant difference in the responses between the noninoculated group and the M. leprae -inoculated group to ConA. In 4/6 of the inoculated animals the response to this mitogen was diminished, depending on the time of the infection. These analyses were carried out over the period of a year at different post-inoculation times; therefore the study of a larger group of animals is needed in order to deduce the role of the infection in nonspecific anergy. Nonspecific anergy has also been reported in mangabey monkeys with disseminated leprosy (7) and in LL patients (1).
The study of the immune response of the armadillo is one of our major interests. This is mainly because the susceptibility of these animals to M. leprae could explain the development of systemic infection. In this work we did not find a complete correlation between proliferation and development of infection. However, recent work extends our observation with the use of lectins as possible markers of lymphocyte subpopulations in these animals (Santos-Argumedo, et al., in preparation). The analysis of cytokines is also another field where we have some preliminary results. These results suggest the role of some of these molecules in the activation of armadillo lymphocytes (Guerra-Infante, et al, in preparation). From these studies we hope to obtain a greater understanding of the development of lepromatous leprosy in humans.
- Laura Rivero-Nava, M.Sc.
Immunology Department
Escuela Nacional de Ciencias Biológicas and Cell Biology Department
Centro de Investigación y Estudios Avanzados, IPN
- Fausto Quesada-Pascual, M.Sc.
Iris Estrada-Garcia, Ph.D.
Sergio Estrada-Parra, Ph.D.
Immunology Department
Escuela Nacional de Ciencias Biológicas
Av. IPN, Prol. Carpio y Plan de Ayala
Mexico, DF 11340, Mexico
- Leopoldo Santos-Argumedo, Ph.D.
Cell Biology Department
Centro de Investigación y Estudios Avanzados, IPN
Apartado Postal 14-740
Mexico, DF 07000, Mexico
Acknowledgment. This work was supported by grants from the Consejo Nacional de Ciencia y Tecnología. México, and the Dirección de Estudios de Post-grado e Investigación, I.P.N., México. The authors hold fellowships from COFAA, EDD and/or SNI, México.
Laura Rivero-Nava was supported during this work by the Consejo Nacional de Ciencia y Tecnología, México, and the Universidad Autónoma de Chihuahua.
REFERENCES
1. BLOOM. B. R. and MEIRA. V. Immunological unresponsiveness in leprosy. Immunol. Rev. 80(1984)5-28.
2. COLSTON, M. J. and LEVY, L. Infection of other experimental animals with Mycobacterium leprae. Int. J. Lepr. 55(1987)896-898.
3. ESTRADA-GARCIA, I., QUESADA-PASCUAL, F., SANTOS-ARGUMEDO, L., FLORES-ROMO, L., ESTRADA-PARRA. S. and BUCHANAN. T. M. The early serodiagnosis of leprosy. I. The use of counterimmuno-electrophoresis and enzyme-linked immunosorbent assay. Rev. Lat. Amer. Microbiol. 26(1984)167-272.
4. HASTINGS, R. C, GILLIS. T. P., KRAHENBUL, J. L. and FRANZBLAU, S. G. Leprosy. Clin. Microbiol. Rev. 1(1988)330-348.
5. KIRCHHEIMER, W. F. and SANCHEZ, R. M. Intraspecies differences of resistance against leprosy in nine-banded armadillos. Lepr. India 53(1981)525-530.
6. KIRCHHEIMER, W. F. and STORRS, E. E. Attempts to establish the armadillo ( Dasypus novemcinctus Linn.) as a model for the study of leprosy. I. Report of lepromatoid leprosy in an experimentally infected armadillo. Int. J. Lepr. 39(1971)693-702.
7. MARTIN, L. N., GORMUS, B. J., WOLF, R. H.. GERONE, P. J., MEYERS, W. M., WALSH. G. P., BINFORD, C. H., HADFIELD, T. L. and SCHAGEL, C. J. Depression of lymphocyte responses to mitogens in mangabeys with disseminated experimental leprosy. Cell. Immunol. 90(1985)115-130.
8. QUESADA-PASCUAL, F., ROJAS-ESPINOSA, O., SANTOS-ARGUMEDO, L. and ESTRADA-PARRA, S. A Mexican armadillo ( Dasypus novemcinctus ) colony for leprosy research. Int. J. Lepr. 55(1987)716-718.
9. SANTOS-ARGUMEDO, L., GUERRA-INFANTE, F, POSADAS-LOPEZ, A., QUESADA-PASCUAL, F. and ESTRADA-PARRA, S. Immune response of armadillos (Dasypus novemcinctus). I. Use of lectins to identify lymphocyte subpopulations and to evaluate cell proliferation. Int. J. Lepr. 63(1995) 546-551.
10. SINGH, N. B., GUPTA, H. P., SRIVASTAVA, A., KAUL. S. M. and SHUKLA, O. P. Antigenic crossreactivity between Mycobacterium habana and Mycobacterium leprae monitored through foot pad enlargement (FPE) in mice. Biol. Mem. 14(1988)199-204.
11. SINGH, N. B., SRIVASTAVA. A., GUPTA, H. P., KUMAR. A. and CHATURVEDI, V. K. Relative crossreactivity of habanin, lepromin and tuberculin in guinea pigs sensitized with homologous and heterologous mycobacteria. Indian J. Lepr. 60(1988)407-412.
12. ULRICH, M., CONVIT, J., CENTENO, M. and RAPETII, M. Immunological characteristics of the armadillo Dasypus sabanicola. Clin. Exp. Immunol. 25(1976)170-176.
Reprint requests to Dr. Santos-Argumedo at the above address or FAX 525-747-7081; e-mail: santos@cell.cinvestav.mx