- Volume 71 , Number 3
- Page: 240–3
A second case of multidrug-resistant Mycobacterium leprae isolated from a japanese patient with relapsed lepromatous leprosy
ABSTRACT
Emergence of drug resistant strains of Mycobacterium leprae was reported soon after the introduction of dapsone (diamino-diphenyl sulphone, DDS) for leprosy treatment (6,10,11). Three cases of multidrug-resistant strains of M. leprae have been reported recently (2, 8, 9,13). In order to prevent multiple drug resistant strains of M. leprae from developing, current leprosy control strategies are based on early detection of cases and treatment with multidrug therapy (MDT) as recommended by the World Health Organization (WHO). We report here the identification of a multidrug-resistant strain of M. leprae from a patient who received inadequate therapy for leprosy. The drug resistant profile of the isolated strain was confirmed by the mouse footpad method and the identification of mutations in genes previously shown to be associated with resistance to each drug was made.RÉSUMÉ
L'émergence de souches de Mycobacterium leprae résistantes aux antibiotiques fut rapportée rapidement après l'introduction de la dapsone (diamino-diphenyl sulphone, DDS) pour le traitement de la lèpre (6, 10, 11). Trois cas de souches de M. leprae résistantes à plusieurs antibiotiques ont été récemment publiés (1, 8, 9, 13). Afin de prévenir l'émergence de souches de M. leprae poly-résistantes, l'approche actuelle du contrôle de la lèpre repose sur la détection précoce des cas et sur une poly-chimiothérapie (PCT) qui suit les recommandations de l'Organisation Mondiale de la Santé (OMS). Nous rapportons ici l'identification d'une souche poly-résistante de M. leprae provenant d'un patient qui a eu un traitement inadéquat contre la lèpre. Le profil de chimiorésistance de la souche isolée fut confirmé par la méthode de la plante des pieds de la souris et l'identification effective des gènes qui ont été démontré comme associés à la chimiorésistance spécifique de chaque antibiotique.RESUMEN
Poco después de la introducción de la dapsona (diamino-difenil sulfona, DDS) para el tratamiento de la lepra aparecieron los primeros reportes sobre la emergencia de cepas de Mycobacterium leprae resistentes a la droga. Recientemente se han publicado tres casos de cepas de M. leprae con resistencia múltiple a las drogas antileprosas. Para prevenir el desarrollo de cepas de M. leprae multidrogo-resistentes las estrategias actuales de control de la lepra se basan en la detección temprana de los casos y el tratamiento con poliquimioterapia (PQT) como se ha recomendado por la Organización Mundial de la Salud (OMS). En este artículo describimos el caso de una cepa de M. leprae multidrogo-resistente derivada de un paciente que recibió un tratamiento inadecuado de la lepra. El perfil de drogo-resistencia de la cepa aislada se confirmó en el modelo de la almohadilla plantar del ratón y por la identificación de mutaciones en los genes asociados con la resistencia a cada droga.A 78-year old, male Japanese patient with leprosy was suspected of harboring drug resistant Mycobacterium leprae as a result of repeated clinical relapses. The patient was diagnosed with lepromatous leprosy (LL) and admitted to the National leprosarium at 31 years of age. The patient was first treated with promin 5 ml/day followed by dapsone 75 to 50 mg/day resulting in regression of skin lesions within one year. The patient's first relapse occurred at 50 years of age. Since the initial relapse, the patient has experienced repeated clinical relapses. During the most recent relapse, a biopsy was taken from a dermal nodule for this study. Since the initial relapse, dapsone, streptomycin, rifampicin, clofazimine, isoniazid, ofloxacin, and prothionamide were administered to treat subsequent relapses. These drugs were administered irregularly as monotherapy or in various combinations, often at doses below recommended levels, and standard multidrug therapy (MDT) was never applied.
The anti-leprosy drug susceptibility of the M. leprae was examined by mouse footpad method (9); the inoculum for footpad studies was prepared from the biopsy sample by propagating M. leprae in the footpads of nude mice. After expansion, M. leprae were tested for susceptibility to various drugs in BALB/c mice. Mice were fed standard pellet mouse chow with or without drugs, as reported previously (9). Experimental groups were fed diets containing one of the following drugs: dapsone, rifampicin, ofloxacin, sparfloxacin, clarithromycin, or clofazimine. Bacillary growth in each footpad was observed individually, according to the standard techniques 30 weeks after inoculation (12). Genomic DNA was prepared from the biopsy material to assess mutations in the rpo, folP and gyrA genes. DNA fragments were amplified that corresponded to regions of rpo (381-bp), folP (350-bp) and gyrA (390-bp) containing mutations, if present, associated with resistance to the rifampin, dapsone, and fluoroquinolones, respectively. Nucleotide sequences of the DNA fragments were determined by direct sequencing and analyzed by the DNASIS program, as described previously (8).
Bacillary growth in footpads treated with rifampicin, ofloxacin, sparfloxacin, and dapsone at concentrations of 0.0001% and 0.001% showed almost the same level of growth as seen in control mice. No bacillary growth was observed in footpads of mice treated with clarithromycin, clofazimine, and dapsone at the concentration of 0.01% (The Figure ). Based on the mouse footpad method, the M. leprae isolate was regarded to be resistant to rifampicin, ofloxacin, sparfloxacin, as well as the low and intermediate levels of dapsone, but susceptible to clarithromycin and clofazimine.
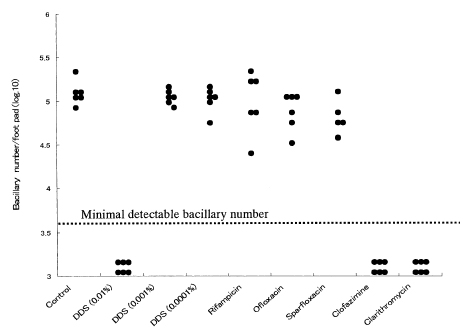
The figure. Bacillary growth in the foot pad of mice administered anti-leprosy drugs.
Sequence analysis of the genes in the M. leprae isolate revealed mutations in folP at codon 55 (CCCTCC), in rpo at codon 531 (TCGTTC), and in gyrA at codon 91 (GCAGTA). These mutations have been shown to be associated with dapsone-, rifampicin-, and quinolone-resistance, respectively in M. leprae.
The results clearly revealed the correlation between relevant mutations and drug resistance to each drug detected by the mouse footpad method. Previous reports have shown that the mutation at codon 55 in folP is a substitution of leucine for proline in high-level dapsone resistant strains (7,8). Therefore, the missense mutation we observed (proline to serine) at codon 55 in the multidrug resistant M. leprae studied suggests resultant dapsone resistance at the intermediate level.
This is the fourth case of M. leprae resistance to multiple drugs used to treat leprosy. Single, or in a few instances, multidrug-resistant M. leprae have been detected among patients who have experienced clinical relapse (1,8,9). This has not been the case for patients treated with current World Health Organization (WHO)-recommended MDT. Standardized MDT regimens were not applied to Japanese patients before 2000 (4). The relapse patient reported in our study had been treated irregularly with several anti-leprosy drugs, suggesting drug resistance was a result of inadequate therapy.
The results of our study strongly support the use of drug resistance monitoring in relapse cases, and demonstrate the utility of drug susceptibility testing by gene mutation analysis. Ideally, gene mutation analysis for drug resistance should be applied when there is suspicion of drug resistance in leprosy. However, this approach is applicable to only three drugs at present. Early detection of drug resistant M. leprae will clarify treatment options and thereby curtail transmission of drug resistant bacilli. Also, we consider it important to begin to establish current levels of drug resistance by surveying leprosy patients for drug resistance using existing molecular tests, as well as developing new methods for detecting drug resistance to current and new drugs that may be useful for treating leprosy.
Acknowledgments. We would like to thank Thomas P. Gillis, National Hansen's Disease Programs, Baton Rouge, LA, USA for his thoughtful ideas for text alterations and consultation regarding this publication. This study was supported by a Health Research Grant of Emerging and Re-emerging Infectious Diseases, Ministry of Health, Labor and Welfare, Government of Japan and US-Japan Cooperative Medical Science Program.
REFERENCES
1. Cambau, E., Bonnafous, P., Perani, E., Sougakoff, W., Ji, B. and Jarlier, V. Molecular detection of rifampin and ofloxacin resistance for patients who experience relapse of multibacillary leprosy. Clin. Infect. Dis. 34(2002)39-45.
2. Cambau, E., Perani, E., Guillemin, I., Jamet, P. and Ji, B. Multidrug-resitance to dapsone, rifampicin, and ofloxacin in Mycobacterium leprae. Lancet. 349(1997)103-104.
3. de Wit, M.Y.L., Faber, W.R., Krieg, S.R., Douglas, J.T., Lucas, S.B., Montreewasuwat, N., Pattyn, S.R.N., Hussain, R., Ponnighaus, J.M., Hartskeel, R.A. and Klaster, P.R. Application of polymerase chain reaction for the detection of Mycobacterium leprae in skin tissue. J. Clin. Microbiol. 29(1991)906-910.
4. Goto, M., Ishida, Y., Gidoh, M., Nagao, E., Namisato, M., Ishii, N. and Ozaki, M. Guideline for the treatment of Hansen's Disease in Japan. (in Japanese). Jpn. J. Lepr. 69(2000)157-177.
5. Grosset, J.H., Guepla-Lauras, C.-C., Bobin, P., Brucker, G., Cartel, J.-L., Constant-Desportes, M., Flageul, B., Frederic, M., Guillaume, J.-C. and Millan, J. Study of 39 documented relapses of multibacillary leprosy after treatment with rifampin. Int. J. Lepr. Other Mycobact. Dis. 57(1989)607-614.
6. Jacobson, R.R. and Hastings, R.C. Rifampin-resistant leprosy. (Letter) Lancet. 2(1976)1304-1305.
7. Kai, M., Matsuoka, M., Nakata, N., Maeda, S., Gidoh, M., Maeda, Y., Hashimoto, K., Kobayashi, K. and Kashiwabara, Y. Diaminodiphenyl sulphone resistance of Mycobacterium leprae due to mutations in the dehydropteroate synthase gene. FEMS Microbiol. Lett. 177(1999)231-235.
8. Maeda, S., Matsuoka, M., Nakata, N., Kai, M., Maeda, Y., Hashimoto, K., Kimura, H., Kobayashi, K. and Kashiwabara, Y. Multidrug resistant Mycobacterium leprae from patients with leprosy. Antmicrob. Agents Chemother. 45(2001)3635-3639.
9. Matsuoka, M., Kashiwabara, Y. and Namisato, Y. A Mycobacterium leprae isolate resistant to dapson, rifampicin, ofloxacine, and sparfloxacin. Int. J. Lepr. Other Mycobact. Dis. 68(2000)452-455.
10. Pearson, J.M.H., Rees, R.J.W. and Waters, M.F.R. Sulphone resistance in leprosy. A review of one hundred proven clinical cases. Lancet. 2(1977)69-72.
11. Pettit, J.H.S. and Rees, R.J.W. Sulphone resistance in leprosy. An experimental and clinical study. Lancet. 2(1964)673-674.
12. Shepard, C.C. and McRae, D.H. A method for counting acid-fast bacteria. Int. J. Lepr. Other Mycobact. Dis. 36(1968)78-82.
13. Shetty, V.P., Uplekar, M.W. and Antia, N.H. Primary resistance to single and multiple drugs in leprosy-a mouse footpad study. Lepr. Rev. 67(1996)280-286.
1. D.V.D., Ph.D., Leprosy Research Center, National Institute of Infectious Diseases. 4-2-1, Aobacho, Higashimurayama, Tokyo, 189-0002, Japan.
2. Ph. D., Leprosy Research Center, National Institute of Infectious Diseases. 4-2-1, Aobacho, Higashimurayama, Tokyo, 189-0002, Japan.
3. M.D., Ph.D., Leprosy Research Center, National Institute of Infectious Diseases. 4-2-1, Aobacho, Higashimurayama, Tokyo, 189-0002, Japan.
4. M.D., Ph.D., Second Department of Pathology, Faculty of Medicine, Kagoshima University, 8-35-1, Sakuragaoka, Kagoshima, 890-8520 Japan.
5. M.D., Ph.D., Division of Surgical Pathology, Kagoshima University Hospital, Faculty of Medicine, Kagoshima University 8-35-1, Sakuragaoka, Kagoshima, 890-8520 Japan.
Reprint request to: Dr. M. Matsuoka at the above address or E-mail: matsuoka@nih.go.jp
Received for publication on 12 December 2002.
Accepted for publication on 13 May 2003.