- Volume 65 , Number 2
- Page: 238–45
Risk of relapse in leprosy after fixed-duration multidrug therapy
ABSTRACT
Between 1986 and 1995, 8307 leprosy patients have completed fixed-duration multidrug therapy (FD-MDT) and were followed annually for possible relapse. The mean relapse rate for multibacillary (MB) leprosy is 0.15/1000 person-years (py) and for paucibacillary (PB) 0.55/1000 py. There is no difference in the relapse rates between patients with or without chemotherapy before FD-MDT. In MB patients, the five relapses occurred between 4 and 7 years; in PB patients, five relapses occurred at 4-5 years after FD-MDT. Six additional PB relapses self-reported 1-4 years after the 5-year surveillance period and were not included in the relapse rates. Most PB patients relapsed into MB due to wrong classification and insufficient therapy. For the known 62 irregular MB patients the cumulative relapse rate is 6.5%.RÉSUMÉ
Entre 1986 et 1995, 8307 malades de la lèpre ont terminé une polychimiothérapie de durée fixe (PCT DF) et ont été suivis chaque année pour des rechutes éventuelles. Le taux moyen de rechute pour la lèpre multibacillaire (MB) est de 0.15/1000 personnes-années et pour la lèpre paucibacillaire de 0.55/1000 personnes-années. II n'y a pas de différence dans les taux de rechute entre les patients qui avaient suivi ou non une chimiothérapie avant la PCT DF. Chez les malades MB, les 5 rechutes sont survenues entre 4 et 7 ans; chez les patients PB, cinq rechutes sont surevnues 4-5 ans après la PCT DF. Six rechutes PB supplémentaires ont été rapportées par les patients eux-mêmes 1 à 4 ans après la période de surveillance de 5 ans, et n'ont pas été inclues dans les taux de rechutes. La plupart des patients PB rechutaient sous une forme MB, suite à une mauvaise classification et à un traitement insuffisant. Pour les 62 patients MB irréguliers connus, le taux cumulé de rechute est de 6.5%.RESUMEN
Entre 1986 y 1995, 8307 pacientes con lepra habían completado su tratamiento con poliquimioterapia de duración fija (I'QT DF) y estaban bajo supervisión anual para detectar posibles recaídas. La tasa promedio de recaída para los pacientes multibacilares (MB) fue de 0.15/1000 persona-años (pa) y para los paucibacilares (PB), de 0.55/1000 pa. No hubieron diferencias en las tasas de recaída entre los pacientes con o sin quimioterapia antes de la PQT DF. En los pacientes MB, las 5 recaídas ocurrieron entre los años 4 y 7; en los pacientes PB, ocurrieron 5 recaídas entre los años 4 y 5 después de la PQT DF. No se incluyeron en las tasas de recaída, seis recaídas PB adicionales autoreportadas, ocurridas entre 1 y 4 años después del periodo de seguimiento de 5 años. La mayoría de los pacientes PB recayeron como MB debido a una clasificación errónea y a la terapia insuficiente. Para los 62 pacientes MB conocidos como irregulares, la tasa acumulativa de recaída fue del 6.5%.Multidrug therapy (MDT) has been implemented world wide since its introduction in 1982 (10) and the relapse rate has been very low (8). However, few publications refer specifically to relapse rates after fixed duration MDT (FD-MDT) (9). In a field study in 1983 involving two independent teams on the therapeutic effect of MDT, ending the regimen at 24-27 months for multibacillary (MB) and 6-10 months for paucibacillary (PB) leprosy, it was noted that the clinical symptoms, bacterial index (BI) and specific infiltration continue to resolve after therapy (5). Encouraged with these preliminary results, and with the aim to enhance the progress of leprosy control, a control program with FD-MDT was launched in 1986 in the most prevalent and the most difficult areas for leprosy control in the country. The present paper presents the findings on the risk of relapse for leprosy patients who have completed FD-MDT within the specified period and have been surveyed annually From 1 to 9 years after therapy.
MATERIALS AND METHODS
The areas chosen for leprosy control with FD-MDT involved 59 counties From seven prefectures of the three southwestern provinces of China, covering 178,758 sq. km, a population of 19 million (1986) and about 10,000 leprosy patients of whom 72% are multibacillary (1986-1995). These prefectures are composed of very rugged mountainous terrain, are populated by more than 20 different ethnic groups, and have the highest leprosy prevalency in the country (0.34-0.74/1000/county, 1985) (The Figure).
The Figure. Map of Yunnan, Guizhou and Sichuan Provinces, China, showing FD-MDT prefectures.
For the present study, a 10-day training course on leprosy control with FD-MDT was held in Xichang, the capital of Liangshan Yi Autonomous Prefecture in November 1985 for 10-12 leprosy workers (LW) From the provincial, prefectural and county levels per province. During this course, methods in case finding, diagnosis (clinical, smear, biopsy), treatment with FD-MDT (MB 24 monthly doses, PB 6 monthly doses of MDT), regularity (MB within 36 and PB within 9 months), management of leprosy/drug reactions, annual clinical and bacteriological surveillance, as well as a recording and reporting system were discussed and standardized. Based upon these lectures and the accompanied practicáis, a manual was prepared and distributed to all LW From the 59 county leprosy control stations (SDCS). All patients who did not complete FD-MDT in the specified period of time are excluded From the present study.
For the prescription of FD-MDT, the patients were designated as PB (10), but in order to study relapses, all patients were classified by the Ridley and Jopling scale clinically and histopathologically.
All new patients, suspected relapses and severe leprosy/drug reactions were first examined by the responsible county LW, checked clinically, bacteriologically and histopathologically, if necessary, by the corresponding specialist From the Provincial Institute of Dermatology (PIOD), usually within 2-3 weeks, and the necessary regimen and/or treatment prescribed. Confirmed relapses were required to complete a special form with all the relevant data before, during and at the end of FD-MDT, plus data at each annual surveillance by the leprologist From PIOD.
Criteria of regularity
The monthly supervised dose was to be taken within 5 days of the scheduled date and the daily self-administered dose was to be taken for at least 20 days per month. The entire course should be completed within 36 months for MB and 9 months for PB.
Criteria of relapse
MB. Criteria for a relapse in MB patients included: a) Insidious appearance of new lesions, enlargement of old lesions, at least 6 months after completing a regular course of FD-MDT; b) an increase in acidfast bacilli (AFB) of >2+ From the same site of the previous examination; c) specific infiltration plus postive AFB in the histopathology of the suspected lesion.
PB. Criteria for a relapse in PB patients was the same as for MB except for AFB.
A 5-day surveillance workshop on cohort analysis was conducted 10 years after the FD-MDT workshop in the prefectural capital of Bijie. Guizhou Province (October 26-30, 1994) for 10 LW responsible for the statistics From the three levels of leprosy administration of each province. This workshop concentrated on the construction of a cohort analysis form, and practicáis for the LW on 30 history records of patients who have completed FD-MDT From Qian-Xi County of Bijie Prefecture.
For the present study, each county leprosy team was required to complete this cohort form for all patients who were on FD-MDT in the county. Data were entered into the databank and analyzed in Beijing.
RESULTS
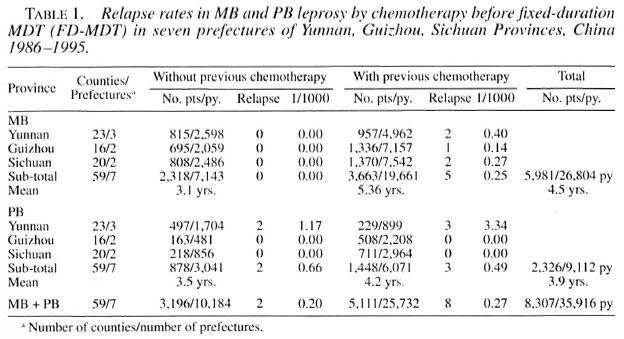
Between 1983 and 1995, 10,047 leprosy patients were put on FD-MDT. Excluding 417 deaths (MB 352, PB 65; 4.2%), 303 lost/moved out (MB 262, PB 41; 3.0%), 74 irregularity (MB 62, PB 12; 0.7%) during FD-MDT and 946 patients still on FD-MDT by the end of 1995, all together 8307 (MB 5981, PB 2326) patients meet the criteria for the present study. Among them, 3196 had never received any antileprosy therapy and 5111 had had some months/years of dapsone or dapsone plus rifampin before FD-MDT (Table 1).
Risk of relapse
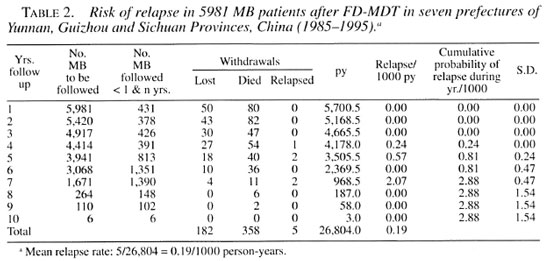
A total of 5981 MB patients had a regular course of FD-MDT, 110 of whom have been followed for 9 years and 6 for 10 years. Since the implementation of FD-MDT in 1986, new patients detected and completing FD-MDT were added to the cohort each year. The period of surveillance therefore varies; with the exemption of irregularity (62), deaths (358) and lost (182), the total follow-up period is 26,804 personyears (py). Five MB relapses (0/7143 py without previous chemotherapy; 5/19,661 py with previous chemotherapy) were detected. These five MB relapses occurred during the fourth (1), fifth (2) and seventh (2) year of surveillance, giving a mean relapse rate of 0.19/1000 py and a cumulative probability of relapse of 2.88/1000 in the seventh year of surveillance (Table 2).
All together 2326 PB patients had completed a regular course of FD-MDT and 1783 PB patients have completed 5 years of surveillance. Five relapses occurred, 4 during year 4 and another during year 5 of follow up, giving a mean relapse rate of 0.55/1000 py excluding death (132) and lost (138). The cumulative probability of relapse is 3.29/1000 in year 5 of surveillance (Table 3).
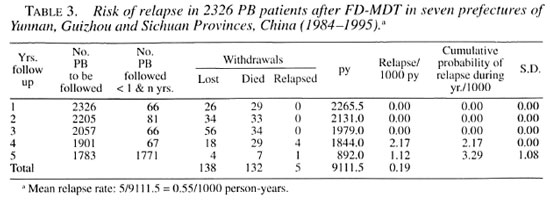
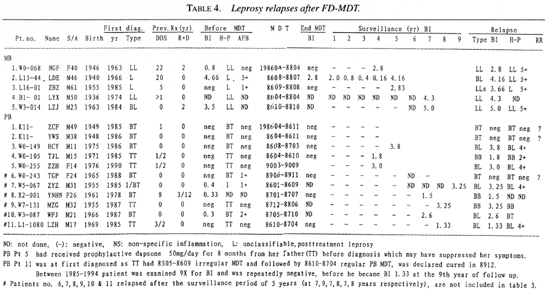
Six PB patients relapsed after year 5 of the surveillance period (3 during year 7, 2 during year 8 and 1 in year 9 after FD-MDT) (Table 4). These relapses were not included in the relapse rates; they were selfreported cases and the surveillance period had terminated at the end of 5 years of follow up.
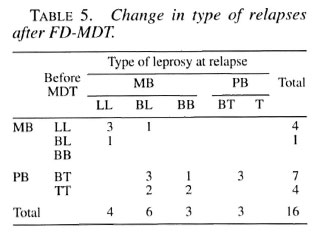
Of the total 11 PB relapses, 7 were originally diagnosed as BT and 4 as TT leprosy (Table 5). Eight PB patients relapsed into MB and 6 of them had >5 lesions and/or >3 nerve involvements at the start of therapy. In the 7 BT relapses, 2 BT (Patients 1 and 2, Table 4) were clinically diagnosed as having a reversal reaction at year 4, and 1 BT patient with a reaction (Patient 6, Table 4) was a self-reported case in year 7 after FD-MDT. All three remained BT in histopathology. A late reversal reaction is likely to be an indication of relapse, and the two BT patients with reversal reaction at year 4 of surveillance were, therefore, counted as relapses.
DISCUSSION
The annual surveillance of the present study follows the rules set forth at the Fourth National Leprosy Conference (Nanchang 1990): MB for 10 years and PB for 5 years after MDT, which is longer than the period required by WHO (7). Using the lifetable method, the mean relapse by personyears of observation for each year after FD-MDT and the cumulative probability of relapse for the successive years of observation are calculated. The latter not only provides the probability of relapse for an individual patient at any year after FD-MDT, but also has the advantage of comparing the therapeutic effect with other treatment regimens at different time intervals.
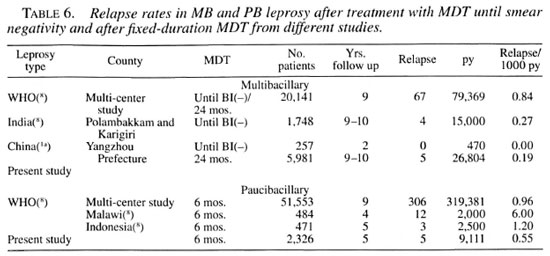
In 3663 MB patients who had received some chemotherapy prior to FD-MDT, 5 relapses occurred: 1 in year 4 and 2 each during years 5 and 7 of follow up, giving a mean relapse rate of 0.25/1000 py. Together with the zero relapse in 2318 MB patients without pevious chemotherapy and 7143 py of follow up, the mean relapse rate is 0.19/1000 py (Table 2). This rate is much lower than the world-wide figure of 0.84/1000 py and is comparable to the relapse rate of 0.27/1000 py From India (Table 6).
Elsewhere China (11 counties in Yangzhou Prefecture, Jiangsu Province), no relapses were noted after treating 347 MB patients until smear negativity, 247 of whom had 470 py of surveillance after therapy (''). Compared with the five relapses in 8307 MB patients in the present study (5/26,804 py = 0.19/1000 py), there is no statistical difference in the relapse rates between MB patients who were treated until smear negativity or who had received only 24 months of MDT, regardless of slit-skin smear results at the time of terminating MDT (X2 = 0.20, p= 1.00).
The difference in relapse rates between MB patients with (5/19,661 py) or without (0/7143 py) chemotherapy before FD-MDT in the present study is also not significant (X2 = 1-74, p = 0.16, Table 1). Prolonged dapsone monotherapy was widely used in China before the introduction of MDT, and primary dapsone resistance was as high as 44% (1). Four out of the five MB relapses were successfully retreated with the MB regimen of FD-MDT (the other pending results of a mouse foot pad study), indicating that MDT can overcome the occurrence of dapsone resistance and its spread.
The mean relapse rate for 878 PB patients without and 1448 PB patients with previous chemotherapy is 0.55/1000 py, which is also lower than the world-wide figure of 0.96/1000 py and figures From Malawi (6.0/1000 py) and Indonesia (1.2/1000 py) (Table 6). There is also no statistical difference between the relapse rates of PB patients with (3/6071 py) or without (2/3041 py) previous chemotherapy in the present study (X2 = 0.13, p = 1.00).
According to WHO statistics, 50% of MB relapses occur within 3 years after MDT and 75% within 6 years; in PB, 50% of the relapses occur within 2½ years and 75% within 5 years after MDT (8).
In the present study, no MB patient relapse occurred within 3 years and all five of the MB relapses occurred 4-7 years after FD-MDT. In PB patients, 4 relapsed in year 4 and 1 relapsed during year 5 of surveillance. There were also 6 late PB relapses, 5 relapsed into MB and 1 cannot be differentiated From reversal reaction.
It is known that a relapse in a PB patient can be difficult to distinguish From a reversal reaction (7). With the recurrence of lesions plus a histopathological picture of BT leprosy and without any evidence for the presence of AFB, three patients (PB patients 1, 2 and 6, Table 5) were counted as PB relapses. From experiences with MB leprosy, reversal reactions occur more often during the first year of FD-MDT and in the first 6 months after FD-MDT, and fall off gradually within 4 years. The BI normally decreases at the rate of about 0.7/year, generally reaching 0 within 5 years after FD-MDT (4). Therefore, it may be assumed that a late reversal reaction is indeed an indication of relapse in PB leprosy. All patients with lepra reactions after FD-MDT, if receiving prednisone, were simultaneously treated with MDT, thus avoiding the lowering of immunity and favoring the multiplication of AFB during corticosteriod therapy. The higher relapse rate in PB patients is mainly due to misclassilication and the prescription of the wrong regimen.
Because of the very difficult field conditions, no attempt in differentiation with a 1-month trial therapy with prednisone was made. All PB relapses were retreated with the MB regimen of FD-MDT.
All five MB relapses were diagnosed as MB before FD-MDT. Of the 11 PB relapses, 8 relapsed into MB and 3 remained PB which cannot be differentiated From reversal reaction. Another three PB relapses were originally AFB positive (PB patients 6, 7 and 10, Table 5) and most PB relapses had >4 lesions and/or >3 nerve involvements before MDT. Moreover, half of the relapses (7/15) were originally diagnosed as BT leprosy, and the PB MDT regimen was administered.
Of the 62 MB patients known to be irregular and excluded From the present study, four relapses/deteriorations (6.5%) were recorded between years 4 and 7 after their scheduled date for FD-MDT. Indeed, poorly implemented MDT is better than no MDT (Yuasa, Y. MDT for all: target oriented leprosy control programme in the 1990s. Paper presented at WHO Consultation on Technical and Operational Aspects of Multidrug Therapy, Male, Maldives 11-15 June 1990).
Based upon the very low irregularity rate (MB: 62/6043 = 1.03%, PB: 12/2338 = 0.51%) and the few late relapses, it may be concluded the FD-MDT is adequate both for MB and PB leprosy, provided that the correct regimen is prescribed and strict regularity of drug intake is adhered to. A correct classification at first diagnosis is of utmost importance. In case of doubt, the MB regimen should be prescribed.
The mean duration of surveillance is only 4.5 years for MB leprosy 3.9 years for PB, and for the sake of further reference, we intend to follow these groups of patients as set at the Nanchang National Leprosy Conference: MB for at least 10 years and PB for at least 5 years after FD-MDT.
As already recommended by WHO, it should be stressed that there is no need to have long-term post-MDT surveillance (s), and there is also no need for any bacteriological surveillance after a regular course of FD-MDT. The BI falls at its own course after adequate therapy regardless of the drug regimen. It is known that MB leprosy may relapse even 20 years after dapsone therapy (5) and 10 years or more after dapsone plus rifampin therapy (2). Patients should be reminded to seek medical care for any recurrence of symptoms, especially during the first year after FD-MDT for leprosy reactions and even years after MDT for possible relapses. The time and manpower thus saved could be devoted to health education, active case finding, disability prevention, integration of leprosy control into the primary health system and health care, in general, at the grassroot level.
Acknowledgment. This investigation received technical and financia] support From the UNDP/World Bank/WHO Special Programme for Research and Training in Tropical Diseases (TDR).
REFERENCES
1. CHEN, J. KWANG, S. Y, HOU, Y. IINI, G. XZHANG, J. L. and TANG, Q. G. Primary dapsone resistance in China. Lepr. Rev 60(1989)263-266.
2. GROSSET, J. HGUELPA-LAURAS, C. C, BODIN, PBRUCKER, GCARTEL, JCONSTANT-DESPORTES, MFLAGEUL, BFREDERIC, MGUILLIAUME, J. C. and MILLAN, J. Study of 39 documented relapses of multibacillary leprosy after treatment with rifampin. Int. J. Lepr. 57(1989)607-614.
3. Li, H.-Y. Problems of leprosy relapse in China. Int. J. Lepr. 61(1993)1-7.
4. Li, H.-Y, HU, L.-FWU, P.-WLUO, J.-S. and Liu, X.-MFixed duration multidrug therapy in multibacillary leprosy. Int. J. Lepr. 65(2)(1997)230-237.
5. Li, H.-Y, Yu, X.-LZHANG, M.-SDUAN, C.-XHUANG, W.-BZHANG, S.-BZHU, K. and MA, J.-F. Short-term multidrug therapy in multibacillary leprosy-review of 80 cases in two provinces of China (1983-1988). Int. J. Lepr. 57(1989)622-627.
6. Li, W. ZYE, G. Y, YANG, Z. MYAN, L. BZHOU, W. QLUO, X. G., TAO, M. BWANG, C. JFENG, BZHU, X. S., Lu, S. QHE, T. S., DING, Z. ZWu, J. Q.. TAO, L. and Ji, F. Five-year observational study on multibacillary leprosy with multidrig therapy. China Lepr. J. 6(1990)61-65.
7. WHO EXPERT COMMITTEE ON LEPROSY. Sixth report. Geneva: World Health Organization, 1988. Tech. Rep. Ser. 768.
8. WHO LEPROSY UNIT. Risk of relapse in leprosy. Indian J. Lepr. 67(1995)13-26.
9. WHO STUDY GROUP. Chemotherapy of leprosy. Geneva: World Health Organization, 1994. Tech. Rep. Ser. 847.
10. WHO STUDY GROUP. Chemotherapy of leprosy for control programmes. Geneva: World Health Organization, 1982. Tech. Rep. Ser. 675.
1. H. Y. Li, M.DM.P.HSenior Researcher; Beijing Tropical Medical Research Institute, 95 Yong An Lu, Beijing 100050, China.
2. L.C. Yuan, Programmer, Beijing Tropical Medical Research Institute, 95 Yong An Lu, Beijing 100050, China.
3. L.F. Hu, M.DDirector; Beijing Tropical Medical Research Institute, 95 Yong An Lu, Beijing 100050, China.
4. Z. Jin, Statistician, Sichuan Provincial Institute of Dermatology, Changdu, Sichuan Province 610031, China.
5. W. B. Huang, M.DDirector; Sichuan Provincial Institute of Dermatology, Changdu, Sichuan Province 610031, China.
6. X. Li, Statistician, Yunnan Provincial Institute of Dermatology, Kunming 650223, China.
7. G.C. Liu, M.DDirector; Yunnan Provincial Institute of Dermatology, Kunming 650223, China.
8. J.L. Li, Statistician, Guizhou Provincial Institute of Dermatology, Guiyang 550001, China.
9. Z.M. Yang, M.DResearch Member, Institute of Cancer, Chinese Academy of Medical Sciences, Beijing 100021, China.
Received for publication on 8 October 1996.
Accepted for publication in revised form on 11 February 1997.