- Volume 65 , Number 2
- Page: 211–6
Comparison of bacillary indexes in slit-skin smears, skin and nerve biopsies; a study f rom Malawi
ABSTRACT
Data analyzed in this paper were collected within the framework of the Lepra Evaluation Project, an epidemiological study of leprosy in Karonga District, northern Malawi. For 212 patients information on the number of skin lesions, slit-skin smear and skin biopsy results were available. Among 61 patients with a single lesion none were slit-skin-smear positive and two had bacilli detected in skin biopsies. In contrast, among 119 patients with four or more lesions 34 (28.6%) versus 59 (49.6%) had bacilli detectable in slit-skin smears or skin biopsies, respectively. In a further 47 patients skin biopsy results could be compared with split-nerve biopsy results. In 20 of 47 patients the bacterial indexes (Bis) were identical in skin and nerve biopsies, while in 26 of 47 patients the Bis were higher in nerve than in skin biopsies. This difference, which is consistent with several other studies in the literature, provides an insight into the pathogenesis of leprosy.RÉSUMÉ
Les données analysées dans cet article ont été récoltées dans le cadre du "Lepra Evaluation Project", une étude épidémiologique de la lèpre dans le District de Karonga, dans le nord du Malawi. L'information sur le nombre de lésions cutanées, les résultats des frottis et des biopsies cutanées étaient disponibles pour 212 patients. Aucun des 61 patients porteurs d'une lésion unique n'était positif à l'examen du frottis cutané, et pour deux on a détecté des bacilles à la biopsie cutanée. Par contre, parmi les 119 patients avec 4 lésions ou plus, 34 (28.6%) et 59 (49.6%) avaient des bacilles détectables respectivement dans les frottis cutanés et à la biopsie. Chez 47 patients supplémentaires, on a pu comparer les résultats d'une biopsie cutanée et d'une biopsie de nerf. Chez 20 des 47 patients, les indices bactériens (IB) étaient identiques dans les biopsies de peau et de nerfs, tandis que chez 26 des 47 patients, les IB étaient plus élevés dans les biopsies de nerfs que dans les biopsies de peau. Cette différence, qui est cohérente avec d'autres études de la littérature, fournit certaines informations quant à la pathogénèse de la lèpre.RESUMEN
Los datos analizados en este trabajo fueron colectados dentro del marco del Provecto de Evaluación de la Lepra, un estúdio epidemiológico de la lepra en el Distrito de Karonga, en el norte de Malawi. Se pudo contar con los datos sobre el número de lesiones en la piei, las baciloscopías en linfa cutânea y los resultados de las biópsias en 212 pacientes. Entre los 61 pacientes con una sola lesión, ninguno tuvo bacilos en linfa cutânea mientras que dos tuvieron bacilos en las biópsias de piei. En contraste, de los 119 pacientes con 4 ó mâs lesiones, 34 (28.6%) tuvieron bacilos en linfa cutanea y 59 (49.6%) tuvieron bacilos en la biopsia de piel. En un grupo adicional de 47 pacientes, los resultados de las biopsias de piel pudieron compararse con los resultados de las biopsias tomadas de los nervios afectados. En 20 de 47 pacientes, los indices bacterianos (Bis) l'ueron idénticos en las biopsias de piel y de nervios, mientras que en 26 de 47 pacientes, los Bis fueron mayores en las biopsias de nervio que en las biopsias de piel. Esta diferencia, la cual es consistente con otros estudios de la literatura, es un aspecto que debe estudiarse a fondo para comprender la patogénesis de la lepra.The presence of acid-fast bacilli (AFB) in slit-skin smears of patients has long been considered as one of the cardinal signs of leprosy, sufficient on its own to establish the diagnosis (4,7). In 1982, the World Health Organization (WHO) recommended the use of multidrug therapy (MDT) as standard treatment for leprosy and introduced an operational classification of leprosy distinguishing between paucibacillary (PB) and multibacillary (MB) forms based on the result of the slit-skin smears (25). This definition was revised in 1988 (24). Several authors have expressed concern about the fact that the decision to treat a patient as PB or MB relies solely on slit-skin smear results since this examination is so dependent upon the availability and quality of slit-skin smear services (3,6), If smear services are not reliable, MB cases could be classified wrongly as PB, thus increasing the risk of relapse due to insufficient treatment. Concern also has been expressed about the difficulty of comparing the effects of MDT between populations and over time, given the changing criteria for classification (3,5).
An increase in case-finding activities within leprosy control programs (LCP) will, in general, lead to an earlier detection of leprosy suspects at a stage when clinical signs are still relatively nonspecific and before the onset of typical nerve damage, increasing the difficulty of the diagnosis. Skin biopsies can help greatly in the diagnosis and classification of leprosy in such individuals (15). Taking biopsies is rarely feasible under routine program conditions, however, in particular if the leprosy control activities are integrated into the general health services. In fact, the vast majority of registered leprosy cases are never biopsied and are diagnosed on clinical examination and on slit-skin smear results alone (5).
Peripheral nerves have long been recognized as preferred sites of Mycobacterium leprae. The bacilli enter and then multiply inside the Schwann cells, inducing a greater or lesser tissue response dependent largely on the immune status of the patient (8,13). There has been increasing interest over the past 20 years in the pathological findings in nerve biopsies (8,13) and the question has arisen whether classifications based on skin and nerve biopsies are comparable (14,28). Several investigators have noted differences in the bacillary load and the histopathological aspects in companion skin and nerve lesions (1,2,9,21,22). These discrepancies have led some authors to question the usefulness and indication of skin and nerve biopsies for the classification and treatment of leprosy (9,12).
To investigate further the nature, frequency and implications of discrepancies between bacteriological findings in skin and nerve lesions, we have compared the results of slit-skin smears, skin biopsies and nerve biopsies in leprosy patients in Karonga District, Malawi.
MATERIALS AND METHODS
Cases for this analysis were ascertained since 1984 within the Lepra Evaluation Project (LEP), a longitudinal study of the risk factors for leprosy in the Karonga District of northern Malawi (17). The entire population was examined at their houses by closely supervised leprosy control assistants (LCA), who had to complete a twopage general examination form for every individual seen. All individuals suspected of having leprosy, and who had no history of previous antileprosy treatment, were examined by a medical officer (JMP). Slitskin smears were taken From anyone with skin lesions which were considered as possibly due to MB leprosy. Two smears were usually taken From the earlobes and two From the lesion(s) and examined at project headquarters. The highest of the four bacterial indexes (Bis) was coded. The percentages of solids, fragments and granules were coded if the BI was 2 or higher. Positive smears of newly discovered leprosy patients were shown to the medical officer by the laboratory technicians. Skin biopsies were obtained From more than 90% of all leprosy suspects newly found in the course of the LEP, and strenuous efforts were made to minimize errors in diagnosis (16). Most biopsies were taken From the most active part of a lesion using a disposable 4-mm punch (10). Split-nerve biopsies were obtained since 1984 a) if the only sign of leprosy was an enlarged peripheral sensory nerve or b) if a suspect agreed to have biopsies taken not only From his skin lesion(s) but also From an enlarged sensory nerve. All biopsy specimens were examined by one of us (SL) and the Bis in the skin and nerve specimens were counted in the same way (oil immersion x1000).
For this analysis two groups of patients have been formed: 1) new ieprosy cases From whom a slit-skin smear and a skin biopsy were taken at the same time. In case of repeated smears and/or biopsies, we chose the first time that these two investigations were performed together. 2) newly diagnosed patients who had a skin and a nerve biopsy taken at about the same time (within 6 weeks). In all of them split-nerve biopsies were taken From an enlarged sensory nerve after the purpose of the investigation had been explained to the patient in detail.
RESULTS
Data were available for the number of skin lesions, slit-skin smear results and skin biopsy results for 212 patients. As shown in Table 1, among 61 patients with a single lesion none were slit-skin smear positive and two had bacilli detected in skin biopsies. In contrast, among 119 patients with four or more lesions 34 (29%) versus 59 (50%) had bacilli detectable in slit-skin smears or skin biopsies, respectively. The latter difference is statistically significant (p <0.01). For the 212 patients, the mean BI was higher in the skin biopsies (0.8) than in the smears (0.5). The difference was statistically significant (t = 4.99; p<0.001).
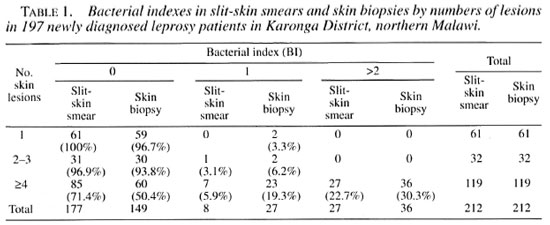
There were 47 patients for whom both skin and nerve biopsy results were available. Their mean age was 39.6 years (42.4 for females; 37.1 for males). Twenty patients had one cutaneous lesion, 5 patients (14%) had two or three lesions and 22 (61%) had four or more lesions. The mean BI was higher in the nerve biopsies (2.4) than in the skin biopsies (1.1) (t = 5.92; p <0.001). The distribution of the Bis in skin and nerve biopsies is shown in Table 2. In 20/47 (43%) patients the Bis were identical in skin and nerve biopsies, in 26 (55%) they were higher in nerve biopsies, and in only one patient (2%) it was higher in the skin biopsy.

The Figure shows the mean Bis in skin and nerve specimens by the histopathological classification based on the examination of the skin biopsies (20). As can be expected, given that high Bis in skin biopsies will lead to a classification of BB to LL leprosy, the difference between nerve and skin biopsy Bis appears greatest for the PB cases [II (indeterminate), TT and BT leprosy].
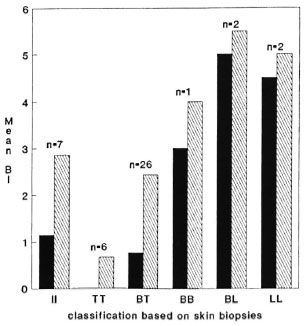
The Figure. Mean bacterial indexes in skin and split-nerve biopsies by histopathological classification based on the skin biopsies From 44 patients in the Karonga District of northern Malawi.  = Skin;
= Skin;  = nerve; n = number of patients in each classification; II = indeterminate leprosy.
= nerve; n = number of patients in each classification; II = indeterminate leprosy.
DISCUSSION
The percentages of slit-skin smears and skin biopsies with a positive BI increased with the number of skin lesions recorded (Table 1). This correlation provides a measure of quality control for slit-skin smear and skin biopsy readings. The histopathologist was not aware of the number of skin lesions found, neither did the laboratory technicians know the number of lesions. The probability of finding bacilli in a patient was greater in the skin biopsies than in the slit-skin smears. One explanation might be that, as suggested by Ridley (19), bacilli in early PB disease are more common in the deeper tissue (particularly nerves) than in the superficial dermal strata, and thus are likely to be excluded From slit-skin smears.
If either the smear- or biopsy-positive BI were to be regarded as evidence of MB leprosy, then 28/177 (15.8%) patients who were suspected on clinical grounds to have MB leprosy would have been "wrongly" classified as PB relying on slit-skin smears only.
Several studies have compared bacteriological loads in skin and nerve biopsies. In a study of 50 leprosy patients undergoing reconstructive surgery in Bombay, Antia, et al. found bacilli in 12% of skin biopsies and in 60% of nerve biopsies (1). Among 30 randomly selected patients attending an outpatient clinic in Chcngalpattu (India), Srinivasan, et al. found that nerve lesions contained more bacilli than skin lesions in six of them (22). In a study of 42 untreated leprosy patients in Nepal, Ridley and Ridley found that the Bis (after correction for the size of granuloma) were generally higher in nerves than in skin biopsies (21). According to Figure 6 in their publication, differences in the Bis were negligible at the two ends of the spectrum but marked for BT to BL leprosy. Negesse, et al. reported a study in which 220 untreated suspected leprosy patients in Ethiopia underwent parallel skin and nerve biopsies. In 46/161 confirmed cases the bacillary load seems to have been considerably higher in nerves than in the skin (11). Similary, in a study of 60 consecutive untreated leprosy patients attending a clinic in Chandigarh (India), the average BI was higher in nerve biopsies than in skin biopsies of the 28 BL patients but not in seven LL leprosy patients (9).
Several hypotheses can be formulated as to why the Bis are higher in nerve than in skin biopsies: a) the difference may simply be due to technical reasons: nerve biopsies are always taken From the best place (an enlarged stretch near the skin) while skin biopsies are usually taken From the most convenient lesion which may not always be the site with the highest density of bacilli; b) M. leprae first settle in nerves and the differences in the Bis simply reflect the longer stay of the bacilli in the nerves than in the skin; c) M. leprae reach nerves and skin at the same time but can survive and multiply more easily in nerves than in skin, perhaps because within nerves they are shielded From the cell-mediated and/or humoral immune system; and d) dead M. leprae are removed more easily From the skin than From the nerves.
It might be possible to refute the first hypothesis by comparing nerve biopsies with several skin biopsies From the same patient. If the Bis in nerves are higher than in all skin specimens, the first hypothesis would become unlikely. The last hypothesis could be tested by comparing percentages of solid-staining (viable) bacilli, which should be higher in the skin than in nerves if dead bacilli are removed more efficiently From the skin than From the nerves. On the other hand, percentages of solids should be similar in the nerves and in the skin if the second or third hypothesis were true. Both hypotheses b) and c) would imply that leprosy does progress From PB to MB within nerves. If, however, bacilli could be found in nerve biopsies but not in several skin biopsies From the same patients, the second hypothesis, that bacilli initially invade nerves, would be the most likely one.
Given that in 32/47 (68%) of biopsied nerves bacilli could be detected, our findings suggest that at least in the absence of reliable slit-skin smear services a leprosy patient should be treated as a MB patient if a definitely enlarged peripheral nerve is found. On the other hand, where slit-skin smear services exist, relapse rates among PB patients are already so low, using the present definition of PB leprosy, that it would appear to be unnecessary to use more stringent criteria for PB leprosy (18).
Acknowledgment. We thank the leprosy patients for their cooperation and the leprosy control assistants for their dedication to the project. The following agencies provided financial support: LEPRA (The British Leprosy Relief Association), ILEP (The International Federation of Anti-Leprosy Organizations) and IMM-LEP (The Immunology of Leprosy component of the WHO/TDR). JMP received a grant From the Robert Cochrane Fund for Leprosy to travel to Bamako to prepare this paper together with CL. We thank the Ministry of Health of the Republic of Malawi for their interest in the project and permission for publication.
REFERENCES
1. ANTIA, N. K. and PANDYA, N. J. Qualitative histology and quantitative bacteriology in various tissues of 50 leprosy patients. Lepr. Rev. 47 (1976) 175-183.
2. ASHOK KUMAR, S. K., REDDY, B. S. N., and RATNAKAR, C. Correlation of skin and nerve histopathology in leprosy. Lepr. Rev. 67 (1996) 119-125.
3. BECX-BLEUMINCK, M. Operational aspects of multidrug therapy. Int. J. Lepr. 57 (1989) 540-551.
4. BRYCESON, A. and PFEALTZGRAFF, R. E. Leprosy. 3rd edtn. Edinburgh: Churchill Livingstone, 1990.
5. FINE, P. E. M. Reflections on the elimination of leprosy. Int. J. Lepr. 60 (1992) 71-80.
6. GEORGIEV, G. D. and MCDOUGALL, A. C. The bacteriological examination of slit-skin smears in leprosy control programmes using multidrug therapy: a plea for radical changes in current operational methodology. Indian J. Lepr. 59 (1987) 373-385.
7. HASTINGS, R. C, ed. Leprosy. Edinburgh: Churchill Livingstone, 1985.
8. JOB, C. K. Nerve damage in leprosy. Int. J. Lepr. 57(1989)532-539.
9. KAUR, S., SHANNA, U. K., BASAK, P. and KAURA, I. Concurrent skin and nerve histology in leprosy and its role in the classification of leprosy. Lepr. Rev. 64(1993) 110-116.
10. MCDOUGALL, A. C, PÓNNIGHAUS, J. M. and FINE, P. E. M. Histopathological examination of skin biopsies From an epidemiological study of leprosy in northern Malawi. Int. J. Lepr. 53 (1987) 88-98.
11. NEGESSE, Y., BEIMNET. K., MIKO, T., WONDIMUS, A. and BERHAN, T. Y. In leprosy the presence of mycobacterium in the nerve is an essential factor in the cycle and spectrum of M. leprae infection. Lepr. Rev. 64(1993) 104-109.
12. NILSEN, R., MENGISTU, G. and REDDY, B. B. The role of nerve biopsy in the diagnosis and management of leprosy. Lepr. Rev. 60 (1989) 28-32.
13. PEARSON, J. M. H. and Ross. W. F. Nerve involvement in leprosy: pathology, differential diagnosis and principles of management Lepr. Rev. 46 (1975) 199-212.
14. PEDLEY, J. C, HARMANN, D. J., WAUDBY, H. and MCDOUGALL, A. C. Leprosy in peripheral nerves: histopathological findings in 119 untreated patients in Nepal. J. Neurol. Neurosurg. Psych. 43 (1980) 198-204.
15. PÖNNIGHAUS, J. M. and FINE, P. E. M. Leprosy in Malawi - 1. Sensitivity and specificity of the diagnosis and the search for risk factors for leprosy. Trans. R. Soc. Trop. Med. Hyg. 82 (1988) 803-809.
16. PÖNNIGHAUS, J. M., FINE, P. E. M. and BLISS, L. Certainty levels in the diagnosis of leprosy. Int. J. Lepr. 53(1987)454-462.
17. PÖNNIGHAUS, J. M., FINE, P. E. M., BLISS, L., SLINEY, I. J., BRADLEY, D. J. and REES, R. J. W. The Lepra Evaluation Project (LEP): an epidemiological study of leprosy in northern Malawi-I. Methods. Lepr. Rev. 58 (1987) 359-375.
18. PÖNNIGHAUS, J. M. and STERNE, J. A. C. Epidemiological aspects of relapses in leprosy. Indian J. Lepr. 67(1995) 35-44.
19. RIDLEY, D. S. The pathogenesis of the early skin lesion in leprosy. J. Pathol. 111 (1973) 191-206.
20. RIDLEY, D. S. and JOPLINO, W. H. Classification of leprosy according to immunity; a five-group system. Int. J. Lepr. 34 (1966) 255-273.
21. RIDLEY, D. S. and RIDLEY, M. J. Classification of nerves is modified by the delayed recognition of M. leprae. Int. J. Lepr. 54 (1986) 596-606.
22. SRINIVASAN, H.. RAO, K. S. and IYER, C. G. S. Discrepancy in the histopathological factors of leprosy lesions in the skin and in peripheral nerve. Lepr. India 54 (1982) 275-283.
23. STONER, G. L. Importance of the nerual predilection of M. leprae in leprosy. Lancet 315 (1979) 994-996.
24. WHO EXPERT COMMITTEE ON LEPROSY. Sixth report. Geneva: World Health Organization, 1988. Tech. Rep. Ser. 768.
25. WHO STUDY GROUP. Chemotherapy of Leprosy for control programmes. Geneva: World Health Organization, 1982. Tech. Rep. Ser. 675.
1. Dr. Med., D.T.M.&H., D.T.P.H., Lepra, P. O. Box 46, Chilumba, Malawi.
2. M.D., D.T.M.&H., M.Sc, Medical Research Council, P. O. Box 273, Fajara, Banjul, The Gambia.
3. F.R.C.P., Professor, UMDS, Guy's and St. Thomas Medical and Dental School, Department of Histopathology, St. Thomas Hospital, Lambeth Palace Road, London SEI 7EH, U.K.
4. V.M.D., Ph.D., Professor; London School of Hygiene and Tropical Medicine, Keppel Street, London WC IE 7HT, U.K.
5. Ph.D., CDEU, London School of Hygiene and Tropical Medicine, Keppel Street, London WC 1E 7HT, U.K.
Reprint requests to Dr. J. M. Ponnighaus, Hautklinik, Vogtlandklinikum Plauen, P.F. 39, 08525 Plauen, Germany.
Received for publication on 16 October 1996.
Accepted for publication in revised form on 15 January 1997.