- Volume 64 , Number 3
- Page: 311–5
Multibacillary nerve histology in clinically diagnosed borderline tuberculoid leprosy patients
ABSTRACT
The classification of leprosy into multi-bacillary (MB) and paucibacillary (PB) patients in almost all clinics is entirely dependent on clinical examination. In a study of 21 patients clinically classified as borderline tuberculoid (BT) and, therefore, belonging to the PB group, skin smears and skin and nerve biopsies were examined. Four patients did not have any histopathological evidence of leprosy. Skin smears showed that 1 patient was positive for acid-fast bacilli (AFB), 2 skin biopsies belonged to the borderline lepromatous (BL) category and showed AFB in their lesions, and AFB were present in 10 nerve biopsies classified as BL. It is possible that reported relapses among PB patients may be in those patients with demonstrable AFB in the lesions, including nerves. A careful follow-up study of this particular group of patients after PB multidrug therapy is suggested to resolve this question.RÉSUMÉ
La classification de la lèpre en patients multibacillaires (MB) et paucibacillaires (PB) dépend dans presque tous les centres de l'examen clinique. Dans une étude de 21 patients cliniquement classifies comme borderline tuberculoides (BT) et donc appartenant au groupe BT, des frottis cutanés et des biopsies cutanées et nerveuses ont été examinés. Quatre patients n'avaient pas le moindre signe histopathologique de lèpre. Les frottis cutanés ont montré qu'un patient était positif pour les bacilles acido- résistants (BAR), deux biopsies cutanées appartenaient à la catégorie borderline lépromateuse (BL) et montraient des BAR dans leurs lésions, et des BAR étaient présents dans dix biopsies de nerfs classifiées BL. 11 est possible que les rechutes rapportées parmi les patients PB pourraient correspondre à ces patients chez, lesquels des BAR ont été mis en évidence dans les lésions, y compris les nerfs. On suggère une étude soigneuse de suivi de ce groupe particulier de patients après polychimiothérapie pour PB pour résoudre cette question.RESUMEN
La clasificación de los pacientes con lepra, como multibacilares (MB) o paucibacilares (PB), se hace en la mayoría de los casos sobre la base del examen clínico. En el presente trabajo se hizo el estudio de la linfa cutánea y de biopsias de piel y nervios, correspondientes a 21 pacientes I'B clasificados clínicamente como tuberculoides subpolares (BT). Se encontró que cuatro pacientes no tuvieron ninguna evidencia histológica de la lepra. El examen de la linfa cutánea mostró que un paciente fue positivo para bacilos ácido resistentes (BAAR). dos biopsias de piel correspondieron a la categoría de lepromatosos subpolares (BL) y mostraron BAAR en sus lesiones, y diez biopsias de nervios clasificados como BL mostraron BAAR. Es posible que las recaídas reportadas entre los pacientes PB pudieran ocurrir principalmente en aquellos pacientes con BAAR demostrables en las lesiones, incluyendo nervios, para analizar esta posibilidad, se sugiere un estudio de seguimiento cuidadoso de este grupo de pacientes PB, después de concluir la poliquimioterapia.The past two decades have seen drastic changes in the time schedule for the treatment of leprosy patients, resulting in short-term antileprosy courses. The World Health Organization (WHO) recommends a standard multidrug therapy (MDT) consisting of 600 mg of rifampin given once a month for 6 months, plus dapsone 100 mg daily for 6 months for paucibacillary (PB) patients and a regimen consisting of rifampin 600 mg once a month, dapsone 100 mg daily and clofazimine 300 mg once a month and 50 mg daily given over a period of at least 2 years for multibacillary (MB) patients (13). MDT coverage has been extensively and successfully used in many areas, but it is obvious that due to these two definite therapeutic regimens the responsibility for categorizing a patient into one or the other is increased. This may be a crucial factor in the ultimate goal of the elimination of leprosy as a public health problem. Classification of leprosy based on the clinical features of characteristic skin lesions still remains the essence of this categorization (4,10). The disparity between the clinical and histological classification across the spectrum of leprosy is known to exist (1,11). Striking differences in the histopathological features of leprosy lesions in the skin and cutaneous nerves have been reported in a few studies (7,12).
This study was done to find out whether such disparity exists in the nerves of clinically diagnosed borderline tuberculoid (BT) skin lesions. Its relevance to the presentday diagnostic and therapeutic trends in leprosy is also discussed in this paper.
MATERIALS AND METHODS
Leprosy patients attending the outpatient department of the Schieffelin Leprosy Research and Training Center in Karigiri, South India, were clinically classified according to the classification described by Ridley and Jopling (10). Among those clinically diagnosed as BT, only those who had one or more enlarged cutaneous nerves and were willing to undergo biopsy of the skin and nerve were selected for the study. The characteristic features of skin lesions that clinically classified a patient as belonging to the BT group included single or multiple, dry, hypopigmented, flat or raised patches of any size with ill-defined or well-defined margins. Modalities of sensation were lost to varying degrees in these patches. Skin smears for acid-fast bacilli (AFB) were done by the slit and scrape method from six routine sites in all patients and selected sites in some patients.
A sliver of nerve from a thickened peripheral cutaneous nerve was biopsied in each of these patients. The most commonly biopsied nerve was the radial cutaneous, followed by the musculocutaneous, ulnar cutaneous and other cutaneous nerves. An elliptical piece of skin was biopsied either from an active skin lesion overlying the thickened nerve or from a similar patch situated away from such a nerve. Biopsies were taken under local anesthesia by the standard Khanolkar technique (6). The skin biopsies were fixed in Formol-Zenker and the nerves in 10% buffered formalin. Serial sections of 5- µ m thickness were stained with hematoxylin and eosin (H&E) for routine study and a modified Fite-Faraco stain for AFB (5).
The skin and nerve lesions were carefully studied histopathologically and classified according to the features described by Job (3) and Ridley (9). The histopathological classification of skin and nerve differ in only some aspects. Caseation is more common in nerve lesions in tuberculoid (TT) leprosy and organisms may be focused in the areas of necrosis. In BT leprosy there are no major differences between skin and nerve lesions. In the nerve in borderline lepromatous (BL) leprosy the lymphocytes are concentrated near the epineurium rather than the entire lesion, and macrophages are relatively few. In lepromatous (LL) leprosy the macrophages are fewer in the nerve lesions, and bacilli are mainly found in Schwann cells. The bacterial index (BI) of the granuloma was assessed for the skin and nerve biopsies separately (9').
RESULTS
Of the clinically diagnosed BT patients, 21 with enlarged cutaneous nerves were studied ( 16 males and 5 females). The duration of the disease ranged from 1 month to 10 years (mean 2 years and 2 months). Eighteen of these patients were untreated and 3 of them had received dapsone monotherapy, 2 for a period of < 2 months and 1 for 3 years' duration. Skin smears were positive for AFB from a selected site in one patient. Skin smears from all other patients were negative.
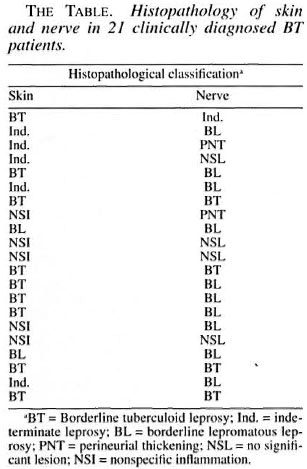
Of the 21 clinically diagnosed BT patients, only 9 of the skin and 4 of the nerve biopsies showed the characteristic granulomatous inflammation of BT histopathology, consisting of poorly organized collections of epithelioid cells admixed with lymphocytes (Fig. 1). Two of the skin and 10 of the nerve biopsies showed BL histology (Figs. 2 and 3). Five skin specimens showed nonspecific chronic inflammation. Two nerve biopsies had only perineurial thickening and four showed no significant lesion (The Table). The BI of two skin biopsies with BL histology were 2+ and in 10 of the nerve biopsies varied from 2 to 4+. All other nerve and skin biopsies were negative for AFB.
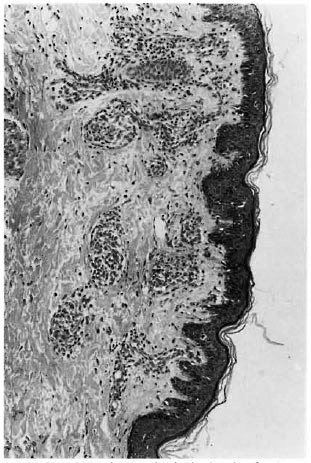
Fig. 1. Photomicrograph of skin showing focal ar-eas of epitheloid granulomas admixed with lympho-cytes in BT leprosy (H&E x100).
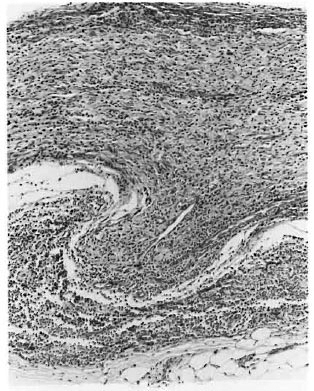
Fig. 2. Photomicrograph of a longitudinal sectionof a nerve with granuloma destroying and protrudingthrough the perineurium in 131. leprosy (H&E. x100).
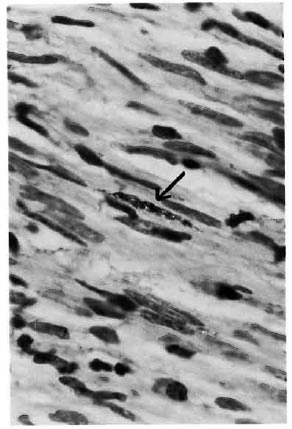
Fig. 3. Photomicrograph showing clump of acid-fast bacilli in a Schwann cell (Fite-Faraco x1000).
DISCUSSION
This study confirms earlier studies describing the disparity between clinical and histopathological diagnosis (1,11). Even with the histopathological examination of both skin and nerve, 4 of the 21 patients (19%) did not have any evidence of leprosy. The importance of the histopathological examination of leprosy lesions in any research study (especially chemotherapcutic trials in PB patients) is clearly brought out here.
Further, this study shows without any doubt the discordance in histopathological appearance of the nerve and skin biopsies from the same patient. A few earlier studies emphasize this important point (7,12). The presence of AFB in skin lesions makes the patient multibacillary and eligible for a 2-year duration of MDT. The absence of AFB makes him/her paucibacillary, qualifying for only 6 months of MDT. In most leprosy clinics, diagnosis is based entirely upon clinical examination, and they are not equipped to do even skin smears. In this study 1 of the 21 BT patients (clinically diagnosed as BT and therefore classified as PB) showed AFB during skin-smear examination. On histopathological study of the skin, two patients had AFB in the macrophages, in dermal nerves and in arrector pili muscle cells of the skin lesions. When cutaneous nerve biopsies were performed, 10 out of 21 (47.6%) were BL patients with AFB in macrophages and Schwann cells. Should all of these patients with demonstrable AFB in the skin/nerve lesions be classified as MB and be given the benefit of a 2-year period of MDT?
One reason pointed out for the comparatively higher relapse rate following MDT in PB leprosy is the possibility of potential MB patients being misdiagnosed as PB leprosy and given the shorter MDT regimen (2,8). The patient who is positive for AFB in skin smears, of course, qualifies for MB MDT. It is imperative to resolve the question of whether M. leprae confined to a few scattered macrophages and dermal or cutaneous nerves makes a patient multibacillary, requiring a longer period of therapy. We intend to follow up the 10 PB patients with AFB in cutaneous nerves and 2 patients with AFB in dermal nerves and an occasional macrophage in the skin lesion to find out the relapse rate among them. A much larger number of PB patients with M. leprae -positive nerve lesions, dermal and cutaneous, will have to be studied and carefully followed up before this question is finally answered.
Acknowledgment. The authors thank Dr. Charles K. Job and Dr. PS.S. Sundar Rao for their comments and advice toward the preparation of this manuscript.
REFERENCES
1. Bhatia, A. S., Katoch, K., Narayanan, R. B., Ramu, G., Mukherjee, A. and Lavania, R. K. Clinical and histopathological correlation in the classification of leprosy. Int. J. Lepr. 61(1993)433-438.
2. Ekambaram, V. and Rao, M. K. Relapse rate in paucibacillary leprosy patients after multidrug therapy in North Arcot District. Indian J. Lepr. 63(1991)34-42.
3. Job, C. K. Pathology of leprosy. In: "Leprosy." 2nd. edn. Hastings, R.C., ed. Edinburgh: Churchill Livingston, 1994, p. 200.
4. Job, C. K. and Chacko, C.J.G. A simplified 6 group classification of leprosy. Lepr. India 54(1981)14-16.
5. Job, C. K. and Chacko, C.J.G. A modification of Fite's stain for demonstration of M. leprae in tissue sections. Indian J. Lepr. 58(1986)17-18.
6. Khanolkar, V. R. Method of taking biopsy tissue for histopathological examination. Lepr. Rev. 22(1951)83-85.
7. Nilsen, R., Mengistu, G. and Reddy, B.B. The role of nerve biopsies in the diagnosis and management of leprosy. Lepr. Rev. 60(1989)28-32.
8. Ramu, G. Duration of MDT for paucibacillary leprosy. Indian J. Lepr. 64(1992)1-7.
9. Ridley, D. S. Skin Biopsy in Leprosy. 3rd edn. Basle: Ciba-Geigy Ltd., 1900.
10. Ridley, D. S. and Jopling, W. H. Classification of leprosy according to immunity; a five-group system. Int. J. Lepr. 34(1966)255-273.
11. Sehgal, V. N., Rege, V. L. and Reys, M. Correlation between clinical and histopathologic classification in leprosy. Int. J. Lepr. 45(1977)278-280.
12. Skinivasan, H. Rao, K. S. and Iyer, C. G. S. Discrepancy in the histopathological features of leprosy lesions in the skin and peripheral nerve. Report of a preliminary study. Lepr. India 54(1982)275-282.
13. Who Expert Committee on Leprosy. Sixth report. Geneva: World Health Organization, 1988. Tech. Rep. Ser. 768.
1. M.B.B.S., MIX. Ag. Head, Department of Histopathology and Experimental Pathology.
2. M.B.B.S., D.C.P., Ag. Head, Department of Immunology and Clinical Pathology.
3. M.B.B.S., Medical Officer. Schieffelin Leprosy Research and Training Center, Karigiri 632 106, North Arcot District. Tamil Nadu, India.
4. M.B.B.S., M.D., Head, Branch of Medicine, Schieffelin Leprosy Research and Training Center, Karigiri 632 106, North Arcot District. Tamil Nadu, India.
Received for publication on 5 December 1995.
Accepted for publication in revised form on 22 April 1996.