- Volume 64 , Number 4
- Page: 428–32
Pterygium in lepromatous leprosy
ABSTRACT
Pterygia f rom the eyes of three lepromatous leprosy patients were histopathologically studied. All of the specimens contained acid-fast bacilli (AFB) and exhibited features of chronic inflammation. In the etio-pathogenesis of the pterygium that occurs in leprosy patients, the chronic inflammation that is a feature of the disease, the involvement of the nerves within the pterygium, the increased exposure to sunlight, dust and wind (especially in patients having lagophthalmos), and the ostrasization by society that necessitates living predominantly outdoor lives, should be taken into account.RÉSUMÉ
Des pterygiums des yeux de trois malades atteints de lèpre lépromateuse ont été étudiés. Tous les specimens contenaient des bacilles acido-résistants (BAR ) et montraient des signes d'inflammation chronique. Dans l'étio-pathogénèse du pterygium qui survient chez les malades de la lèpre, l'inflamation chronique qui est une caractéristique de la maladie, l'atteinte des nerfs à l'intérieur du pterygium, l'augmentation de l'exposition à la lumière du soleil, la poussière et le vent (particulièrement chez les patients qui ont une lagophtalmie), et le rejet par la société qui force à mener une vie principalement à l'extérieur devraient être pris en considération.RESUMEN
Se estudió la histopatología de los ojos con pterigia de 3 pacientes lepromatosos. Todos los especímenes tuvieron bacilos ácido resistentes y mostraron signos tic inflamación crónica. En la etiopatogénesis de la pte rigia que aparece en los pacientes con lepra debe tomarse en cuenta la participación de factores tales como la inflamación crónica característica de la enfermedad, la afección de nervios dentro del pterigio, y la exposición prolongada a la luz solar, al polvo y al viento, especialmente en los pacientes con lagoftalmos.Pterygium is a fairly common but not necessarily innocuous lesion of the eye, and is characterized by a triangular mass of thickened conjunctiva that encroaches on the corneal limbus in the interpalpebral fissure, more often on the nasal side than on the temporal. Histologically there is evidence of degeneration of the collagen fibers resulting in the accumulation of granular amorphous material in the subepithelial tissues. It is often found in those working outdoors who are exposed to wind, sunlight and dust. We report here the clinico-pathological features of three lepromatous patients with pterygium associated with lepromatous granuloma of the conjunctiva.
CASE REPORT 1
A 64-year-old female presented with a history of progressive decrease in vision in both eyes and irritation in the right eye for a period of 6 months. In 1974 she was diagnosed as having indeterminate leprosy and treatment was started with dapsone 100 mg daily. After a month the patient left her residence, went out of the leprosy control area and was lost to follow up until April of 1990, when she came to the hospital for a hand ulcer, at which time dapsone was again started. After a month, she once again was lost to follow up until February 1996.
On examination the patient had partial anesthesia of both the upper and lower limbs. There was also partial loss of sensation over the face. The ulnar, posterior tibial and the common peroneal (lateral popliteal) nerves were thickened bilaterally. There was alopecia, clawing of both hands and a left foot drop. Routine clinical-pathological investigations were within normal limits.
Skin smears for acid-fast bacilli (AFB) in March of 1996 showed a bacterial index (BI) of 4+. A biopsy of a representative skin patch from the right buttock confirmed the patient as having lepromatous leprosy.
An ocular examination was done, and the right eye vision was 2/60. There was madarosis and lagophthalmos. A thick, fleshy medial pterygium extending and crossing the pupillary area was present. A corneal opacity about 2 mm in diameter was present at the 7 o'clock position of the cornea. A leash of blood vessels was seen entering it from the pterygium. Decreased corneal sensation and an age-related cataract were also present. The left eye had a vision of 6/60 with madarosis, decreased Cornell sensation and an age-related cataract. The pterygium in the right eye was excised and sent for histopathological examination.
CASE REPORT 2
A 50-year-old male clinically classified as lepromatous leprosy 30 years ago, and who had had 8 years of monotherapy followed by multidrug therapy as advocated by WHO (9) 3 years previously, presented with a mass in the left eye. On examination the patient was found to be a typical case of lepromatous leprosy with characteristic diffuse fine infiltration and wrinkling of almost the entire skin of the body. There was bilateral madarosis. In the left eye there were old keratic precipitates, the pupil was irregular with posterior synechia and the iris was atrophic. Skin smears showed a BI of 3.5+. The left eye had a pterygium-like lesion encroaching the nasal cornea. This was excised and sent for histopathological study.
CASE REPORT 3
A 65-year-old male diagnosed clinically as lepromatous leprosy of unknown duration, and who had had 10 years of dapsone monotherapy, reported with pain in the left eye. On examination there were extensive grossly infiltrated lepromatous lesions all over the body, face and extremities. There was lagophthalmos of the left eye with conjunctival congestion and keratic precipitates in the cornea. Skin smears were 4.5+. There was a pterygium-like lesion in the nasal side of the cornea. This was excised and sent for histopathological examination.
HISTOPATHOLOGY
The tissues excised from the eye were fixed in 10% buffered neutral formalin for over 48 hr and processed for paraffin sections. Five- µ m sections were cut; one section was stained with hematoxylin and eosin (H&E), one for AFB according to a modified Fite's stain (3) and a third for Weigert's elastic tissue stain (6).
All three biopsies had essentially similar histopathological appearances. The tissue was lined by stratified squamous epithelium showing some areas of hyperplasia. The subepithelial connective tissue was locally infiltrated with fairly large collections of inflammatory cells composed of lymphocytes, plasma cells, macrophages and occasional neutrophils (Fig. 1). There was also marked tissue edema and scattered inflammatory cells throughout the section. Hyaline degeneration of the subepithelial collagen bundles was present (Fig. 2). In addition, in one specimen there was a subepithelial clump of pink granular material. In two of the specimens there was proliferation of nerve bundles and peri- and intraneurial infiltration by chronic inflammatory cells (Fig. 3). One specimen showed well marked hypertrophy of goblet cells forming gland-like structures in the lining epithelium (Fig. 4).
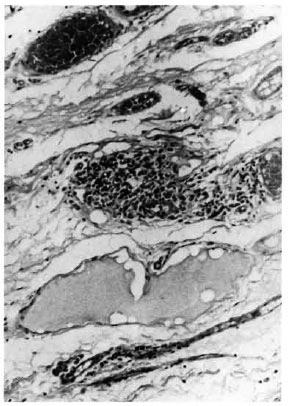
Fig. 1 . Photomicrograph showing focal collection of inflammatory cells composed of lymphocytes, plasma cells, and macrophages. There is also little tis-sue edema (H&E x 100).

Fig . 2. Photomicrograph showing hyaline degeneration of subepithelial collagen tissue (H&E x 100).
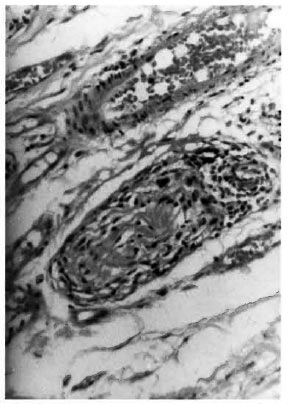
Fig . 3. Photomicrograph showing cross-section of a nerve bundle with peri- and intraneurial inflammation (H&E x 100).
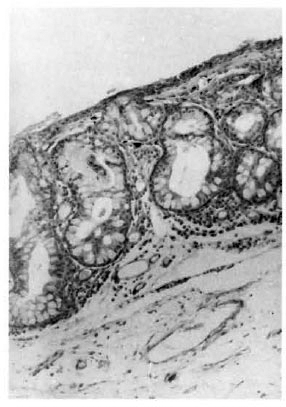
Fig. 4. Photomicrograph showing hypertrophied goblet cells forming gland-like structures (H&E x100).
Acid-fast stain showed bacilli in macrophages (Fig. 5) in all three specimens, and in one the AFB were found inside Schwann cells (Fig. 6), endothelial cells and fibroblasts. A section stained for elastic fibers failed to show any fragmented granular elastic tissue.
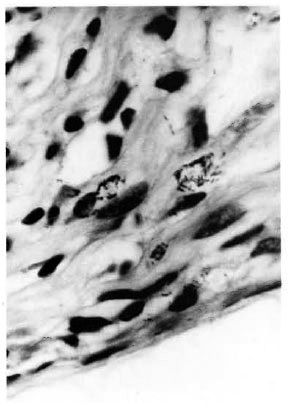
Fig . 5. Photomicrograph showing clumps of M.leprae inside macrophages (modified File's stain x 1000).
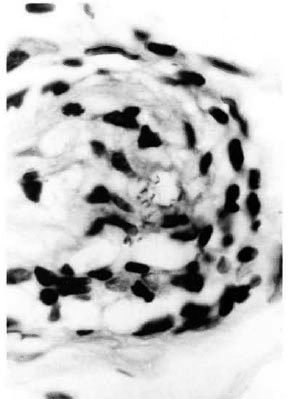
Fig . 6. Photomicrograph showing section of nerve bundle with Schwann cells containing M. leprae (modified Fite's stain x 1000).
DISCUSSION
Pterygium is still an enigma in ophthalmology (2). Although the wing-shaped fleshy growths have been known to physicians for thousands of years, very little is known about the pathogenesis of this condition. It has been accepted that environmental factors are responsible for pterygium development and that ultraviolet light has the most important environmental influence (7). The way in which ultraviolet light interacts with the limbus to produce a pterygium is not clearly understood. The histopathology is nonspecific with hyaline degeneration and a low-grade inflammatory reaction that does little to suggest an underlying cause.
Nerves, when found within the pterygium, have not been accorded any significance in the pathogenesis, but in a study of 93 tissue specimens of pterygia, investigators (6) found both myelinated and unmyelinated axons in one specimen and suggested that there may be a neurotropic component to pterygium pathogenesis. In our study, two of the three pterygial tissues showed prominent nerves in the H&E sections. There were many AFB present inside Schwann cells. The histopathology of these pterygial tissues also showed intense chronic inflammation related to the AFB within the tissue. The inflammatory reactions present in a pterygium are usually of a low grade (6). The leprosy-related granulomatous reactions seen within the pterygium may be a contributory factor in the pathogenesis of pterygium in these patients.
Another contributing factor to the development of pterygium in leprosy patients could be the increased levels of ultraviolet light that these patients are likely to be subjected to as a result of ostracization from society which makes them lead predominantly outdoor lives. Lagophthalmos, which is a well known ocular complication in leprosy, may be an additional contributing factor when present. Two of the patients from whom the pterygia were removed had lagophthalmos. The eye in lagophthalmos is exposed to more dust and ultraviolet light.
One of the earlier studies done on the occurrence of pterygium in leprosy patients (3) reported that it occurred twice as commonly in leprosy patients as in the general population, but another study (4) stated that the distribution was equal in both leprosy patients and in the general population. Both of these studies were done four decades ago, and it would be interesting to see the prevalence of pterygium in leprosy patients living in the present time in which antileprosy chemotherapy has played a more effective role in the control of leprosy.
Acid-fast staining of the pterygia showed AFB that were solid staining, indicating that viable bacilli had infiltrated the tissue. It is likely that the bacteria entered the tissue through its blood vessels as evinced by the presence of AFB in the endothelium of the blood vessels within the pterygium.
Elastic staining was negative in all three of our specimens, but this is not an unusual finding since actinic elastosis is reportedly uncommon in pterygia found in India (1) unlike in some parts of the world where almost all pterygia are associated with elastotic degeneration.
REFERENCES
1. ANSARI, M. W., RAMI, A. H. S. and SHUKI.A, B. R. Pseudoelastic nature of pterygium. Br. J. Ophthalmol. 54 (1970) 453-460.
2. COSTER, D. Pterygium-an ophthalmic enigma. Br. J. Ophthalmol. 79 (1995) 304-305.
3. GARCIA DE AZEVEDO, J. Piterigio na lepra ocular. Arq. Mineiros Leprol. 4 (1944) 85. Abstracted in Int. J. Lepr. 16(1948) 123.
4. HOLMES, W. J. Changing concepts in ocular leprosy. Trans. Pacific Coast Oto-Ophthal. Soc. 35 (1954) 51-56. Abstracted in Int. J. Lepr. 23 (1955)473.
5. JOB, C. K. and CHACKO, C. J. G. A modification of Fite's Stain for demonstration of M. leprae in tissue sections. Indian J. Lepr. 58 (1986) 17-18.
6. KARUKONDA, S. R. K., THOMPSON, H. W., BEUERMAN, R. W., LAM, D. S. E., WILSON, R., CHEW, S. J. and STE;INE;MANN, T. L. Cell cycle kinetics in pterygium at three latitudes. Br. J. Ophthalmol. 79 (1995)313-317.
7. MORAN, D. J. and HOLLOWS, F. C. Pterygium and ultraviolet radiation: a positive correlation. Br. J. Ophthalmol. 68 (1984) 343-346.
8. SHEEHAN, D. C. and HRAPCHAK, B. B. Theory and Practice of Histotechnology. 2nd edn. Columbus: Battle Press, 1980, pp. 194.'
9. WHO STUDY GROUP. Chemotherapy of leprosy for control programmes. Geneva: World Health Organization, 1982. Tech. Rep. Ser. 675.
1. M.S., Head, Department of Ophthalmology; Schieffelin Leprosy Research and Training Center, Karigiri 632106, North Arcot District, Tamil Nadu, India.
2. M.D., Acting Head, Depart-ment of Histopathology; Schieffelin Leprosy Research and Training Center, Karigiri 632106, North Arcot District, Tamil Nadu, India.
3. M.B.B.S., Medical Officer, Branch of Epidemiology; Schieffelin Leprosy Research and Training Center, Karigiri 632106, North Arcot District, Tamil Nadu, India.
4. M.D., F.A.M.S., E. R.C. Path., Emeritus Scientist, Schieffelin Leprosy Research and Training Center, Karigiri 632106, North Arcot District, Tamil Nadu, India.
5. F.R.C.S. (Ophthal.), Head, Department of Ophthalmology, Purulia Leprosy Hospital, Bonilla,West Bengal 723101, India.
Received for publication on 24 June 1996.
Accepted for publication in revised form on 20 Sep-tember 1996.